1. Introduction
China has vast saline alkali land resources, covering an area of 9.91×107 hm2,1 widely distributed in northern regions of China, the vast majority are in a state of barrenness, with extremely low source utilization efficiency.2 Meanwhile, with the improvement of living standards, people’s demand for aquatic products is increasing, however, the water resources that can be used for aquaculture are very limited.3 Therefore, the development and utilization of saline alkali water resources for aquaculture is of great significance. Saline alkali water has the characteristics of high salinity and pH values, which makes it difficult for most aquatic organisms to adapt, and the yield of aquaculture is unstable. Exploring the potential resource advantages of saline alkali water and using it for aquaculture can help solve the problem of scarce freshwater resources in fisheries.4 However, currently, there are not many suitable aquaculture objects for saline alkali water cultivation. Therefore, it is necessary to explore more excellent aquaculture varieties capable of adapting to saline alkali environments. The largemouth black bass (Micropterus salmoides) is a popular fish that is suitable for commercial aquaculture. Now, it has become one of the main freshwater aquaculture species in China.5,6 Compared with other conventional fish such as four major Chinese carps, M. salmoides can efficiently regulate ion regulation and ammonia nitrogen excretion, thus adapting to saline and alkaline environment. Therefore, M. salmoides has strong adaptability to saline and alkaline environment, and is a freshwater economic species suitable for saline and alkaline water aquaculture.7 Currently, M. salmoides has been successfully cultured in some saline alkali water areas such as Ningxia and Tianjin, but its adaptation range and mechanism to saline alkali conditions are still unclear.7
Salinity is one of the most important abiotic factors affecting the growth and survival of fish.8,9 The salinity range of 0.5-0.8% is approximately the critical salinity for the distribution of aquatic organisms. This salinity is the lower limit of salinity for most marine organisms and the upper limit of salt tolerance for most freshwater organisms.10 In the study of Siganus gutatus, it was found that the fish had a strong tolerance to low salt. When the salinity changes rapidly, S. gutatus could clear excess ROS in the body by regulating its own antioxidant enzyme activity.11 In the study of Scatophagus argus, a high salinity aquaculture environment could cause oxidative stress in the fish’s body.12 Studies have also shown that an increase in salinity could reduce the survival rates of M. salmoides and Triplophysa dalaica.13,14 Experiments in water bodies with different salinities had shown that salinities of 0.4% and 0.8% could reduce the growth performance of M. salmoides.15 However, currently, there are few reports on the effect of salinity on antioxidant and metabolism-related enzyme activities, tissue structure, and changes in the gut microbiota of M. salmoides.
pH is also one of the important indicators affecting the growth and survival of fish. When the water environment is slightly alkaline (pH>9.0), it could cause direct corrosiveness to fish, causing serious damage to the surface of the fish and tissues such as gills.16 Studies have shown that the northwest region of China is mostly a saline alkali area, with a pH generally ranging to around 8.4.17 Wang 's (2023) research showed that the optimal pH for the growth of M. salmoides is 8.0, and under this condition, the specific growth and survival rates of M. salmoides were also the highest.16
Environmental factors such as salinity and pH stress can cause fish to be unable to maintain normal physiological states, leading to changes in antioxidant levels and immune indicators to a certain extent, or unable to recover to normal levels, and affecting organ functions such as the liver and gills, as well as the balance of gut microbiota. This seriously affected the normal growth, development, feeding, and other behaviors of fish, ultimately resulting in decreased disease resistance, slow growth, and even large-scale mortality.11 Besides, it is more meaningful to combine salinity and pH for research, firstly because the saline alkali water area has both high salinity and pH.4 Combining analysis is more important for the promotion and breeding of M. salmoides in the northwest region. Secondly, studies have shown that the impact of pH on the growth and survival of fish is often accompanied by the synergistic effects of other factors, such as an increase in salinity under high pH conditions, which may further enhance the damage to fish bodies.16 Thirdly, there are numerous water environmental factors that can affect the normal growth and survival of fish. However, in saline alkali water environments, salinity and pH may be the two most important factors, and their main effects on fish bodies may vary, requiring further exploration. Therefore, in the future, more in-depth research is needed on the impact of relevant environmental factors on fish, exploring the mechanisms of the combined effects of different environmental factors on fish. Besides, the changes in antioxidant and metabolism-related enzyme activities, tissue structure, and changes in the gut microbiota should be used as an important basis for water quality testing and fish health. An early warning plan that combines environmental factor stress response to fish with water quality monitoring should be established, providing an important scientific basis for the healthy development of fishery aquaculture and aquaculture in saline alkali environments.
In view of this, in the context of improving aquaculture management, we explored the effects of salinity and pH stress on M. salmoides. We established three stress groups to compare the antioxidant and metabolism-related enzyme activities, tissue structure, and changes in the gut microbiota of each group. The aim of the study was to explore the effects of salinity and pH on the comprehensive characteristics of M. salmoides. The results may provide guidance for the breeding programs for stress tolerance and management practices in saline alkaline water bodies of M. salmoides in the future.
2. Materials and methods
2.1. Experimental animals and study conditions
The experiments were conducted at the Zhejiang Institute of Freshwater Fisheries, Huzhou, China. M. salmoides, a domesticated variety, after having been raised from hatchling to adult stage in an outdoor enclosure, were evenly distributed and physically healthy, and randomly selected for the experiment. A total of two hundred and sixteen fish were used. The average body weight of the selected fish was 548.30±58.04 g and the body length of 26.09±0.68 cm. The experimental container was a polypropylene plastic breeding tank containing 100 L of water. The water temperature, ammonia nitrogen, nitrite, and pH were maintained at 26.0±1.0°C, at 0.1-0.3 mg/L, 0.01-0.1 mg/L, and 7.3-10.0. The tank was equipped with continuous inflation to maintain a dissolved oxygen concentration of 6.5-7.5 mg/L.
2.2. Methods
2.2.1. Salinity and pH stress test
Based on the results of the acute toxicity pre-test, there was 100% mortality rate at 24 h occurred in M. salmoides in water with pH was 10.5 or the salinity was 1.0% (Table 1). Therefore, three experimental concentration gradients were set up, namely group 1 (control group, salinity 0%, pH 7.3, at 96 hours and 192 hours, samples were taken as 1-1 and 1-2 groups), group 2 (0.75% salinity and 9.0 pH stress group, at 96 hours and 192 hours, samples were taken as 2-1 and 2-2 groups), and group 3 (10.0 pH and 0.5% salinity stress group, at 96 hours and 192 hours, samples were taken as 3-1 and 3-2 groups), with 3 parallel replicates set for each concentration. In the experiment, the salinity of water was adjusted to the corresponding levels for each group, and the fluctuation of salinity in water was maintained within ± 0.5 ‰ under corresponding conditions using sea crystals. Moreover, an industrial digital salinity meter was used to detect the salinity of each group. Besides, the pH of water was adjusted to the corresponding levels for each group, and the fluctuation of pH in water was maintained within ± 0.1 under corresponding conditions using 0.01 mol/L HCl and 0.01 mol/L NaOH standard solutions. Moreover, a digital pH meter was used to detect the pH of each group.
A total of two hundred and sixteen fish were used. Each parallel box contained 100 L of the test solution, and twelve fish were placed in the test box for salinity and pH experiments. M. salmoides did not die at 192 h in each group. Six fish were collected from each group after 96 h, and another six fish were collected after 192 h. The fish were anesthetized using 100 mg/L MS-222, and dissected on an ice tray to promptly excise liver, intestinal, and gill tissues. The method of euthanizing the fish followed the ethical standards for the treatment of experimental animals. The liver and intestinal samples were stored at -80 ℃ for measuring enzyme activity and microbiota, and the liver and gill samples were placed in Bonn’s solution for tissue sectioning and observation. This study was approved by the Ethics Committee of Laboratory Animal Center of Zhejiang Institute of Freshwater Fisheries.
2.2.2. Preparation of tissue samples
At 96 h and 192 h, six fish were randomly selected from each treatment tank and killed. The liver and gills were immediately excised, and part of the liver and gills tissue were preserved in liquid nitrogen for subsequent preparation for the enzyme analysis. The rest of the liver and gills tissue were prepared for slides. The liver and gills were fixed with Bonn’s solution for 24 h, dehydrated step by step with ethanol, made transparent with xylene, embedded in paraffin, sliced with a 5 µm microtome, stained with hematoxylin eosin, and sealed with neutral gum. The intestinal tissue was cut open with sterile scissors, and the contents were gently scraped with a dissecting knife and placed in liquid nitrogen and transferred it to a -80°C freezer for later use in microbiome analysis. The samples prepared were examined under a biological microscope and photographed.
2.2.3. Measuring antioxidant and immune related enzyme activity
Excised livers that had been stored in liquid nitrogen followed by storage at ultra-low temperature (-80°C) were used to prepare homogenate samples for enzyme analysis. The tissue homogenate was prepared by adding 9 mL of physiological saline per gram of the sample tissue, grinding the tissue, transferring it to a 2 mL centrifuge tube, and centrifuging it at 1006.2 ×g g at 4 °C for 10 min. The resulting supernatant was divided into small portions, and stored temporarily in a refrigerator at 4°C. The activity and content of SOD, MDA, ACP, and AKP were measured using reagent kits. Three replicates per enzyme assay in each group (group 1-1, 1-2, 2-1, 2-2, 3-1, 3-2). The relevant operations were performed according to the manufacturer’s instructions of the reagent kit, and all reagent kits used were from the same batch.
2.2.4. Analysis of differences in composition and diversity of gut microbiota
The prepared intestinal tissue (see above, 2.2.2 Preparation of tissue samples) was used to investigate the microbiota of the fish from each of the treatment groups. Total DNA was extracted from each group, using the E.Z.N.A. Fecal DNA Kit (D4015). DNA extraction quality was detected by 1% agarose gel electrophoresis. PCR amplification was performed using universal primers 341F: 5 ‘- CCTACGGGNGGCWGCAG-3’ and 805R: 5 ‘- GATACHVGGGTATCTAATCC-3’ for the V3-V4 region. PCR products were detected by 2% agarose gel electrophoresis. PCR amplification program: Pre-denature at 98 ℃ for 30 seconds, 32 cycles (denature at 98 ℃ for 10 seconds, anneal at 54 ℃ for 30 seconds, and extend at 72 ℃ for 45 seconds); finally extend at 72 ℃ for 10 minutes. Purification of PCR products using Ampure XT beads and quantification using Qubit.
Sample analysis included Illumina PE250 sequencing and bioinformatics analyses. Genomic DNA extracted from the sample was subjected to Chain Reaction (PCR) amplification, fluorescence quantification, Illumina PE250 library construction, and Illumina PE250 sequencing. The PE reads obtained from Illumina PE250 sequencing were first concatenated based on the overlap relationship, and sequence quality was controlled and filtered. After distinguishing between the samples, Operational Taxonomic Unit (OTU) clustering and species taxonomy analyses were performed. Based on the OTU clustering analysis results, various diversity index analyses were performed on the OTUs, as well as the detection of the sequencing depth. Based on taxonomic information, a statistical analysis of community structure was conducted at various taxonomic levels.
2.3. Statistical analysis
All data were presented as mean ± SD and utilized to assess the normal distribution and uniformity. After arcsine transformation, the data obtained for each experiment were analyzed by analysis of variance (ANOVA) using Excel and SPSS software to determine the differences between groups. Tukey’s multiple comparison post-hoc test was performed when significance was detected. Statistical significance was set at P < 0.05.
3. Results
3.1. Toxicity test of salinity and pH on M. salmoides
The results showed that during the breeding period, none of the fish in each group die. However, in groups 2 and 3, we observed that during the initial stage of stress, some fish began to show signs of stress: rapid breathing, an increased frequency of gill opening and closing, and hyperactive behavior. As the level of stress increased (especially at 192 h), some fish frequently rolled or collided and darted about before resting at the bottom.
3.2. Effects of salinity and pH stress on liver antioxidant and immune related indicators
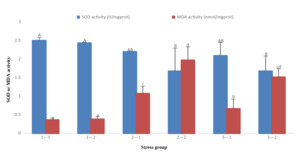
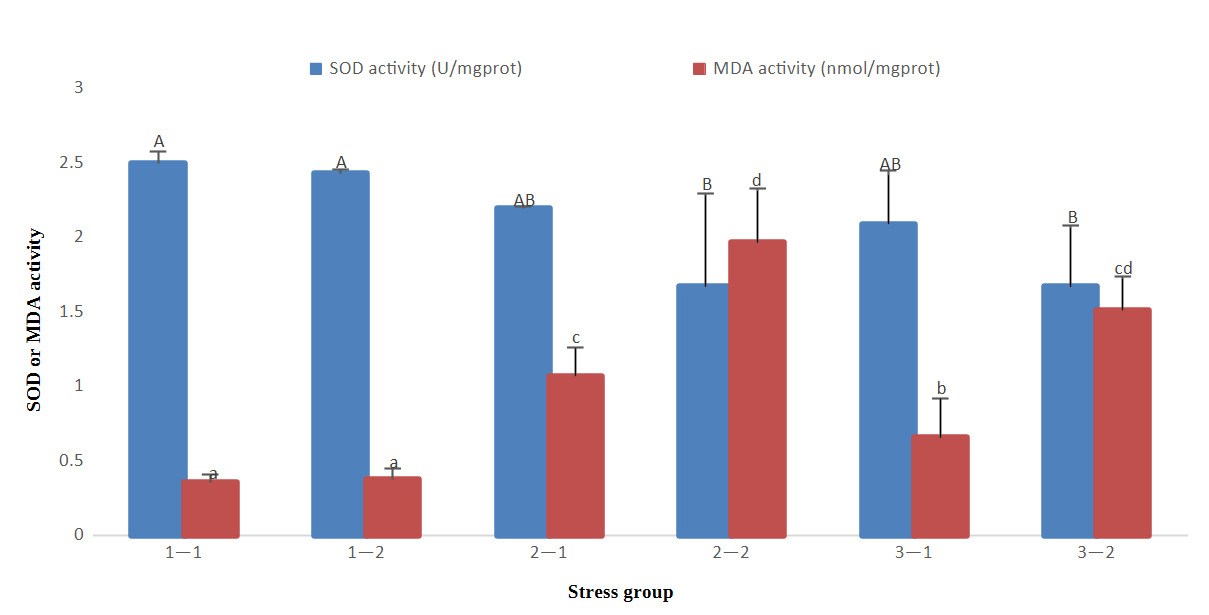
The results of antioxidant indicators showed that with the prolongation time of stress, SOD gradually decreased in groups 2 and 3, and MDA gradually increased in groups 2 and 3, but there was no significant decrease or increase in group 1. At 96 h of treatment, there was no significant difference in SOD activity in the three groups (P>0.05), however, MDA levels in group 1 were significantly lower than those in the other groups (P<0.05) and group 2 were significantly higher than those in the other groups (P<0.05). At 192 h of treatment, SOD activity in groups 2 and 3 was significantly lower than that in group 1 (P<0.05); however, MDA activity in groups 2 and 3 was significantly higher than that in group 1 (P<0.05). Group 2 had high MDA activity at 96 h and 192 h (Fig. 1).
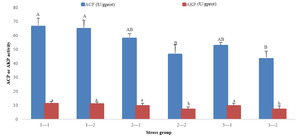
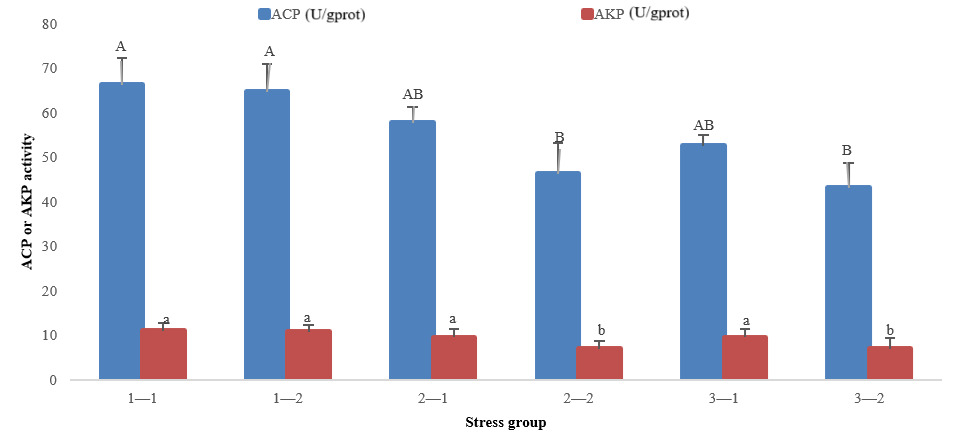
The immune-related indicators showed that with the prolongation time of stress, ACP and AKP activities decreased in groups 2 and 3, but there was no significant decrease or increase in group 1. At 96 h of treatment, there was no significant difference in ACP and AKP activities in three groups (P>0.05). At 192 h of treatment, ACP and AKP activities in groups 2 and 3 were significantly lower than those in group 1 (P<0.05) (Fig. 2).
In conclusion, the SOD, ACP, and AKP activities of groups 2 and 3 at 192 h were significantly lower than that of group 1 at 96 h and 192 h, whereas the MDA index of groups 2 and 3 was significantly higher than that of group 1 at 96 h and 192 h.
3.3. Effects of salinity and pH stress on the structure of liver and gill tissues
3.3.1. Histological observation of liver tissue
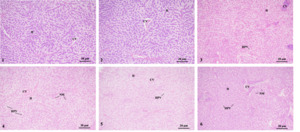
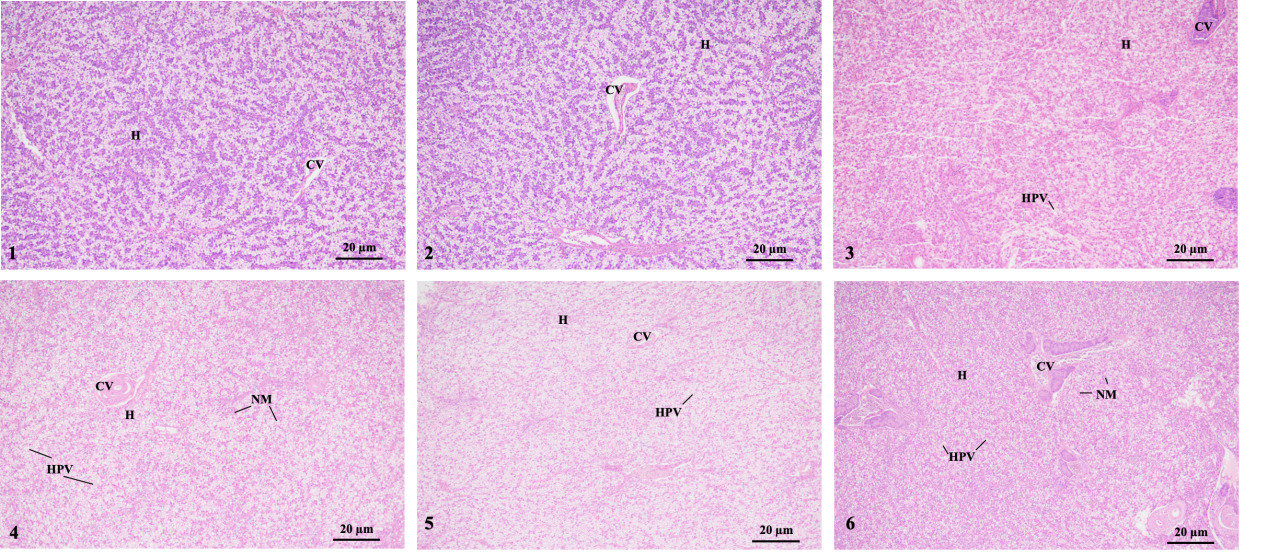
The histology showed that liver cells in group 1 (1-1 and 1-2 groups) were arranged neatly and had clear cell contours. However, in groups 2-1 and 3-1, some liver cells appeared large areas of vacuolization. Moreover, groups 2-2 and 3-2 had dilated sinusoids, swollen liver cells, severe vacuolization, and a severe nuclear shift. That was, with an increase in stress time, the swelling, vacuolization, and nuclear shift of liver cells gradually worsened (Fig. 3).
3.3.2 Histological observation of gills
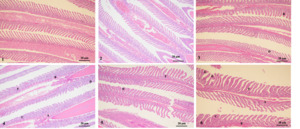
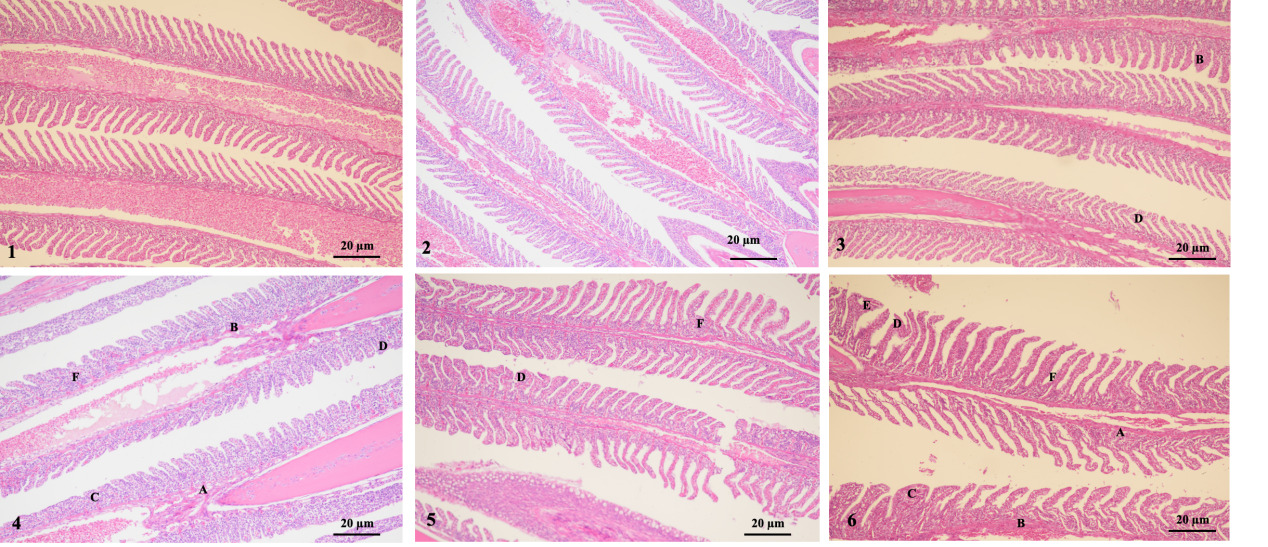
The results showed that group 1 (1-1 and 1-2 groups) had intact gill filaments, neatly arranged gill filaments, and no obvious swelling, congestion, fusion, curling, or peeling was found. However, group 2-1 showed significant hyperemia and filament curling. Group 3-1 showed hyperemia and hyperplasia. Moreover, groups 2-2 and 3-2 exhibited severe epithelial swelling, congestion, gill filament fusion, gill filament-layered curling, and hyperplasia. Therefore, as the stress duration increased, the swelling and congestion of the gill epithelium, gill fusion, layered curling, and proliferation of gill filaments gradually became more severe (Fig. 4).
3.4. Effects of salinity and pH stress on gut microbiota
3.4.1. Analysis of microbial community composition
The results showed that the gut microbiota of M. salmoides mainly included Fusobacteriota, Proteobacteria, Actinobacteria, and Firmicutes. Among them, Fusobacteriota, Proteobacteria, and Actinobacterota were more abundant in group 1-1. Firmicutes, Proteobacteria, and Actinobacterota were more abundant in group 1-2. Fusobacteriota were the most abundant phyla in group 2-1. Actinobacterota and Fusobacteriota were more abundant in group 2-2. Fusobacteriota, Proteobacteria and Firmicutes were more abundant in group 3. In other words, at the phylum level, after high salinity and moderate pH stress, the abundance of the phyla Proteobacteria decreased, while the abundance of the phyla Fusobacteriota increased at 96 h, however, the abundance of the phyla Fusobacteriota decreased, while the abundance of the phyla Actinobacteria increased at 192 h. Besides, after high pH and moderate salinity stress, the abundance of Actinobacteria increased at 96 h; however, it decreased at 192 h. At 192 h, except for Fusobacteriota, Proteobacteria, and Firmicutes, there is almost no other phyla. In conclusion, after high salinity and moderate pH stress, Proteobacteria decreased at first, and then increased; Fusobacteriota increased at first and then decreased. After high pH and moderate salinity stress, Actinobacteria increased at first, and then decreased. Besides, the main phyla were becoming increasingly concentrated.
The genera with the highest abundance at the genus level were Cetobacterium, Dietzia, Aeromonas, Mycoplasma, and Candidatus-Bacilloplasma. Among these, the above five genera were all abundant genera in group 1-1. Candidatus-Bacilloplasma was more abundant in group 1-2 at the genus level. The genera with the highest abundances in group 2-1 were Dietzia and Cetobacterium. The most abundant genera in group 2-2 were Aeromonas and Cetobacterium. Group 3-1 and 3-2 exhibited a higher abundance of Cetobacterium. In other words, at the genus level, after high salinity and moderate pH stress, the abundance of Aeromonas, Mycoplasma, and Candidatus-Bacilloplasma decreased, while the abundance of Dietzia increased at 96 h, however, the abundance of Dietzia and Candidatus-Bacilloplasma decreased, while the abundance of Cetobacterium and Aeromonasc increased at 192 h. After high pH and moderate salinity stress, the abundance of Dietzia, Aeromonas, and Candidatus-Bacilloplasma decreased, while the abundance of Cetobacterium increased. In conclusion, after high salinity and moderate pH stress, Aeromonas, Mycoplasma, and Candidatus-Bacilloplasma decreased at first, and then increased; Dietzia increased at first, and then decreased. After high pH and moderate salinity stress, Aeromonas decreased at first, and then increased; besides, the abundance of Cetobacterium was always high (Fig. 5, Table 2).
3.4.2. Analysis of microbial diversity index
Groups 1-1, 1-2, 2-1, 2-2. 3-1, and 3-2 contained an average of 85,723, 85,717, 91,086, 92,017, 89,084, and 90,530 mean sequences, respectively. The ACE and Chao1 indices of groups 1-1, 1-2, and 2-1 were significantly higher than the corresponding values in groups 2-2, 3-1, and 3-2 (P<0.05). The Shannon indices of groups 1-1 and 1-2 were significantly higher than the corresponding values in the other four groups (P<0.05), whereas the Simpson index was significantly lower than that of the other four groups (P<0.05). The ACE and Chao1 indices of groups 3-1 and 3-2 were significantly lower than the corresponding values in the other four groups (P<0.05). The Shannon index of group 3 1 was significantly lower than the corresponding values in the other five groups (P<0.05), whereas the Simpson index was significantly higher than that of the other five groups (P<0.05) (Table 3).
4. Discussion
4.1. Acute stress of salinity and pH in M. salmoides
In the present study, there was 100% mortality rate at 24 h occurred in M. salmoides in water with pH was 10.5 or the salinity was 1.0%. However, M. salmoides did not die at 192 h in water with pH was 9.0-10.0 and the salinity was 0.5-0.75%. Studies have shown that salinity levels of 0.4% and 0.8% could reduce the growth performance of freshwater M. salmoides.15 There were also studies indicating that an increase in salinity could reduce the survival rate of M. salmoides. It was speculated that this might be due to the fact that M. salmoides, as freshwater fish, had weaker self-regulation abilities in certain salinity water bodies, and their water salt balance was easily disrupted.13 In addition, studies had shown that the optimal pH for the growth and survival of M. salmoides was 8.0. When the water environment was slightly alkaline (pH>9.0), it could cause direct corrosiveness to the fish, causing serious damage to the surface of the fish and tissues such as gills. It could also stimulate the gills to secrete a large amount of mucus, which condensed in the gills and made it difficult for the fish to breathe and suffocate. If the pH of the water environment caused the surface mucosa of the fish’s skin to dissolve, it would further lead to the fish losing its ability to control water in order to maintain osmotic pressure.16 It was also observed that during the initial stage of stress, M. salmoides showed symptoms of severe stress (detailed in Section 3.1), especially stress at 192 h. It was speculated that the increase in salinity and pH in the water might lead to the disruption of the fish’s water salt balance, causing damage to the surface, gills, and internal organs of the fish, which affected the fish’s osmotic pressure balance and movement control ability.
4.2. Antioxidant and immune related enzyme activity indicators of salinity and pH in M. salmoides
The results showed that the SOD, ACP, and AKP activities of groups 2 and 3 at 192 h were significantly lower than that of group 1 at 96h and 192 h, whereas the MDA index of groups 2 and 3 was significantly higher than that of group 1 at 96 h and 192 h.
Stress can increase the reactive oxygen species concentration in fish, further inducing oxidative stress and causing oxidative damage.18,19 SOD, as a free radical scavenger, has significant effects in responding to acute situations in redox reactions and signal transmission processes, maintaining metabolic balance in the body through a series of reaction processes.20 Studies have shown that salinity had a significant impact on antioxidant indicators such as SOD in M. salmoides.13 In the study of S. gutatus, it was found that when the salinity of water was 0.5%, there was no significant difference in SOD enzyme activity between the control group. Until the salinity of water up to 1.0-2.0%, there was still a certain degree of increase in SOD activity.11 Another study showed that the antioxidant enzyme activity of M. salmoides was normal at a salinity of 0.5%, and it did not cause oxidative damage.16 The results of this study showed that under pH 9.0-10.0 and salinity range of 0.5-0.75%, there was no significant difference in SOD activity between the experimental groups and the control group at 96 h, however, the SOD activities of experimental groups were significantly lower than the control group at 192 h, which was slightly different from the previous research results. This might be related to the synergistic effect of pH. Meanwhile, MDA content reflected the rate and intensity of lipid peroxidation in the body, and was an important indicator of the body’s potential antioxidant capacity, when MDA was high, the body would suffer oxidative damage.13 Yang (2020) found that in juvenile M. salmoides, if excessive reactive oxygen species and MDA in liver cells were not cleared in a timely manner, the cells would undergo oxidative damage, leading to a decrease in antioxidant capacity.21
The pH of the water environment also has an impact on farmed fish, and most farmed fish are suitable for survival in neutral and weakly alkaline environments. Inappropriate high pH in the water environment can dissolve the mucous membranes on the skin surface of fish, further causing them to lose their ability to control water in order to maintain osmotic pressure.22 Study of Wang (2023) had shown that the optimal pH for the growth of M. salmoides was 8.0, and its growth rate and survival rate were higher than those of the group of pH was 8.5.16 The results of this study showed that under salinity of 0.5-0.75% and pH range of 9.0-10.0, there was no significant difference in SOD content between the groups at 96 h, however, the stress experimental group was significantly lower than the control group at 192 h, while MDA content was significantly higher than the control group.
Based on the changes in SOD and MDA content in this study, it was speculated that under pH 9.0-10.0, the salinity was 0.5-0.75% for a certain period of time, and the body might suffer oxidative damage.
ACP and AKP are important lysosomal enzymes involved in non-specific immunity.13 Therefore, changes in the activities of these two enzymes may, to some extent, indicate the immune status of the body.23 The results were similar with respect to SOD, indicating that with the prolongation of stress time, the activity of ACP and AKP decreased. Dong et al. (2021) studied the effects of water salinities (0.3-0.9%) on the growth and immune-related enzyme (ACP, AKP) activity of juvenile M. salmoides (28.46±0.1g). The results showed that there was no significant difference in the activity of these two enzymes at different salinities, but when the salinity increased to 0.9%, the oxidative damage to the liver of M. salmoides intensified, and the growth performance and survival rate significantly decreased.13 The results of this study were slightly different from the above results, and it was speculated that it might be related to the size of the fish body and the synergistic effect of salinity and pH.
4.3. Histological observation of liver and gills of salinity and pH in M. salmoides
The results showed that stress with salinity and pH caused histological damage to the liver and gills. The liver is an organ for detoxification and metabolism.24 The histological observation of the liver in this experiment showed that with an increase in stress time, the swelling, vacuolization, and nuclear shift of liver cells gradually worsened. Wang et al. (2025) studied the effects of salinity on the liver and gill tissue structure of Paramisgurnus dabryanus and found that high salinity and moderate high pH and moderate salinity stress could cause certain damage to the liver and gill tissues of P. dabryanus. Under high salinity stress (1.2%), liver tissue cells exhibited phenomena such as vacuolization and nuclear shift and speculated that vacuolization of liver cells might be caused by an imbalance between the rate of substance synthesis and the rate of release into the circulatory system.25 Jiang et al. (2007) also reached a similar conclusion in their study of Chalcalburnus chalcoides aralensis.26 Tao et al. (2016) investigated the effects of high pH stress on the liver tissue structure of Procambarus clarii. The results showed that with prolonged high pH stress, the basement membrane of the hepatic pancreatic tubules was damaged, the number and volume of vacuoles in the tubules increased, the nucleus shifted, and the number of liver cells decreased.27 The results of the above experiment were similar to those of this study, where liver cells exhibited phenomena such as vacuolization and nuclear shift. It was speculated that when the concentration of external stress factors, such as salinity and pH, exceeded a certain amount or time, the normal tissue structure of the liver might be damaged, which might affect normal physiological functions.
Gills are important organs for osmotic pressure regulation in bony fish, and their morphological structure can undergo significant changes in response to complex external environments.28 In the present study, the histological observations of the gills were similar to those of the liver. As the stress time increased, epithelial swelling and congestion of gill filaments, gill filament fusion, gill filament layer curling, and proliferation gradually became more severe. Study have shown that excessive salinity (1.2%) could damage the gill tissue of juvenile Luciobarbus capito, causing shedding of gill epithelial cells, congestion, and swelling. Besides, it was found that under high salt stress, the gill tissue of the large scale loach became shorter, the distance between gill plates increased, and the epithelial cells of gill plates dissolved.29 Similar research results also include study on Lateolabrax maculatus.30 Liu et al. (2025) studied the tissue damage of M. salmoides under different salinity stress. The results showed that tissue necrosis and cell shedding occurred in the gills at salinities of 1.5% and 1.0%. When the salinity was 0.5%, the gill tissue was not damaged. Among them, the pathological features of gill shedding and intestinal villus rupture observed at a salinity of 1.5% suggested that tissue repair ability might be inhibited by high salinity.31 Wang et al. (2013) studied the effect of different pH values on the microstructure of gills of Scophthalmus maximus. The results showed that in the group of pH was 8.3, detachment of gill epithelial cells was observed, along with damage caused by defense stress responses such as proliferation of gill filament epithelial cells, partial keratinization, and edema of gill respiratory epithelial cells. In contrast, in the group of pH was 8.8, keratinization of gill epithelial cells and shedding of gill epithelial cells of Paramisgurnus dabryanus were more severe.25 The results of the above experiments were similar to those of the present study.
4.4. Composition and diversity of gut microbiota of salinity and pH in M. salmoides
The results showed that at the phylum level, as stress persisted, Proteobacteria first decreased and then increased; however, Fusobacteriota first increased and then decreased.
The gut microbiota plays an important role in the host immune response and stability of the internal environment.32 Changes in the aquatic environment can trigger stress responses in animals, affecting the gut microbiota structure.33 Huang et al. (2024) found that the abundance of gut microbiota in M. salmoides at the phylum level was the highest in Firmicutes, Proteobacteria, Fusobacteriota, and Actinobacteria.34 This was consistent with the results of this experiment; however, Proteobacteria, Fusobacteriota, and Actinobacteria were in a significant dynamic state during stress. Proteobacteria contains many pathogenic bacteria, including Escherichia coli and Salmonella, if these bacteria dominate the gut microbiome, the body is prone to diseases.35,36 Fusobacteriota is less harmful and plays a beneficial role in the gut microbiota. It may not only produce beneficial metabolites such as pyruvic acid and butyric acid but also participate in the maintenance of gut microbiota homeostasis and metabolism.37,38 Actinobacteria have beneficial bacteria such as bifidobacteria, but there are also many members that are pathogenic.39 Therefore, as stress persisted, harmful bacteria such as Proteobacteria first decreased and then increased, while harmful or beneficial bacteria such as Fusobacteriota first increased and then decreased. It was speculated that as the stress persisted, the imbalance of gut microbiota and the increase of harmful bacteria would lead to an increased risk of fish diseases and immune dysfunction.
In addition, the results of this study indicated that the genera with the highest abundances were Cetobacterium, Aeromonas, Mycoplasma, Candidatus_Bacilloplasma, and Dietzia. Among them, the last four genera were in a significant dynamic state during stress. Cetobacterium plays an important role in the glucose and lipid metabolism of fish and can enhance their immunity and promote intestinal health.40 Aeromonas are widely distributed in aquatic environments. Most bacteria in this genus have been reported to have strong pathogenicity in fish.41 Mycoplasma is a highly polymorphic gram-negative pathogenic microorganism that lacks a cell wall. Many Mycoplasma are not pathogenic, but some species may also cause cell damage.42 Candidatus_Bacilloplasma is a novel lineage of Mollicutes that is associated with the hindgut wall of the terrestrial isopod Porcellio scaber. This lineage was positioned as a sister group to the clade comprising the Spiroplasma and Mycoplasma groups.43 Dietzia belongs to the phylum Actinobacteria and has strong salt and alkali tolerance. Verified through pan genetic analysis and physiological experiments that this genus of bacteria has broad-spectrum alkane degradation ability and good high salt and alkali tolerance.44 Further research has found that it contains a novel sodium ion/proton (Na+/H+) antiporter protein that plays a major role in regulating intracellular Na+ and H+ homeostasis. Sodium ion/proton antiporter (Multiple resistance and pH-related antiporter) complex is a type of multi-subunit transmembrane protein that catalyzes the efflux of monovalent cations such as Na+, K+, and Li+ and the intracellular transport of protons.45 There has been relatively little research on this bacterium, and further exploration is required in the future. Therefore, overall, as stress persisted, Aeromonas, Mycoplasma, and Candidatus-Bacilloplasma decreased at first, and then increased; Dietzia increased at first and then decreased. It was speculated that as the stress persisted, the increase of harmful bacteria as Aeromonas and Mycoplasma, would lead to an increased risk of fish diseases and immune dysfunction. Besides, Dietzia, as a bacterial genus with strong salt and alkali tolerance, might play an important role in regulating the body’s homeostasis during stress periods, the first increase in its abundance indicated that it responded to stress to reduced the impact of salinity and pH on the body. As stress time increased, the decrease in its abundance might indicate the disruption of its microbial community homeostasis.
The results showed that stress with salinity and pH led to a decrease in gut microbiota diversity. The microbiota diversity decreased with increasing stress time, especially group 3-2 (high pH and moderate salinity stress group at 192 h), which had the lowest microbiota diversity. Previous studies have shown that a decrease in diversity may lead to a decrease in the functional stability of bacterial communities.46 Zhao et al. (2024) investigated the response of gut microbiota of Gymnocypris przewalskii to different high salinity and moderate high pH and moderate salinity stresses and found that high salinity (salinity≥1.5%) stress would inhibit the growth of gut microbiota and dominant microbiota of G. przewalskii and had a significant impact.47 Wei et al. (2024) found that ammonia stress for 48 h significantly reduced the alpha and beta diversity of the gut microbiota in M. salmoides, leading to decreased stress tolerance and susceptibility to intestinal dysfunction.48 This study was similar to previous research in that stress led to a decrease in gut microbiota diversity.
Therefore, the stress with 9.0-10.0 pH and 0.5-0.75% salinity caused changes in enzyme activity, damage to the liver and gills, and alterations in the gut microbiota in M. salmoides. In actual aquaculture, the changes of antioxidant and immune-related indicators, liver and gill tissues, and gut microbiota are closely related to the salinity or pH stress, which can become a part of breeding programs for stress tolerance and is helpful for guiding management practices in saline alkaline water bodies of M. salmoides.
5. Conclusion
In summary, the stress with 9.0-10.0 pH and 0.5-0.75% salinity caused changes in antioxidant and immune-related enzyme activity indicators, histological damage to the liver and gills, and alterations in the main gut microbiota and diversity in M. salmoides. In actual aquaculture production, close attention should be paid to the condition of fish after stress to prevent disease or damage to the body or organs. Future research should evaluate the long-term stress responses of salinity and pH on M. salmoides adaptability, reproduction, and population dynamics, as well as their application in selective breeding of M. salmoides.
Acknowledgments
This work was financially supported by Basic Public Welfare Research Program of Zhejiang Province (LGN22C190024), Major Science and Technology Programs for Agriculture (Aquaculture) New Variety Selection of Zhejiang Province (2021C02069-2) and Zhejiang Provincial Financial Special Project (2025CZZX02).
Authors’ Contribution
Conceptualization: Shun Cheng, Chao Zhu; Methodology: Mei-li Chi, Chao Zhu; Formal analysis and investigation: Wen-ping Jiang; Writing - original draft preparation: Shun Cheng; Writing - review and editing: Fei Li, Shi-li Liu; Funding acquisition: Fei Li, Shun Cheng, Shi-li Liu; Resources: Jian-bo Zheng, Mei-li Chi, Jian-mei Xin; Supervision: Fei Li, Shi-li Liu.
Competing of Interest – COPE
No competing interests were disclosed.
Ethical Conduct Approval – IACUC
This study was approved by the Ethics Committee of Laboratory Animal Center of Zhejiang Institute of Freshwater Fisheries. The animal protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of the Zhejiang Institute of Freshwater Fisheries. The animal study was reviewed and approved according to the guidelines of the Animal Experiment Committee, Zhejiang Institute of Freshwater Fishery (ZIFF20240302).
Informed Consent Statement
All authors and institutions have confirmed this manuscript for publication.
Data Availability Statement
All are available upon reasonable request.

_and_genus_level_(below_figure)_for_ea.png)
_and_genus_level_(below_figure)_for_ea.png)