1. Introduction
The rapid development of intensive aquaculture methods and the reduction in fishmeal supply have led to a sharp rise in fishmeal prices.1 Adding nutrients such as carbohydrates and lipids to feed is a method to enhance the protein-sparing effect of aquaculture feed.2 Compared with lipids, carbohydrates have better advantages in terms of price and availability.3,4 Appropriate supplementation of carbohydrates in feed can not only improve economic efficiency but also reduce ammonia nitrogen emissions, thereby achieving the effect of reducing water pollution.5 To produce cost-effective aquaculture feed, commercial feeds often contain excess carbohydrates.6 However, carnivorous fish have a limited ability to utilize carbohydrates, and long-term consumption of high-carbohydrate diets can not only lead to liver metabolic disorders in fish but also impair intestinal health.7,8 Nevertheless, the specific mechanisms by which high-carbohydrate diets induce intestinal damage in carnivorous fish remain to be fully elucidated. Moreover, effective functional additives to mitigate such damage remain lacking, which significantly hinders the rational application of dietary carbohydrates in carnivorous fish nutrition.
The intestine is a complex digestive organ that plays a crucial role in the body’s digestion, absorption, and metabolism.9However, nutritional levels are the main factor affecting Intestine health.10In studies on Nile tilapia (Oreochromis niloticus), the high-carbohydrate (HC) diets showed increased expression levels of inflammatory cytokines.11 In studies on largemouth bass(Micropterus salmoides), the HC diets altered the gut microbial composition, with a decrease in the abundance of beneficial bacteria and an increase in the abundance of harmful bacteria.7 In studies on blunt Snout Bream (Megalobrama amblycephala), the HC diets caused abnormal intestinal morphology, increased intestinal permeability, and suppressed intestinal immunity.12It is noteworthy that the potential role of the functional additives of the HC diets in mitigating the adverse effects of the HC diets on fish has garnered growing attention. Betaine has attracted significant attention due to its potential application value derived from its core functions, such as anti-inflammatory and barrier protection properties. Betaine is a type of quaternary ammonium alkaloid, also known as trimethylglycine, Betaine was first discovered by the German chemist Scheible in sugar beets (Beta vulgaris).13,14As is well known, betaine is commonly used as a feed additive to maintain the intestine health of terrestrial animals.15For example: Betaine has been shown to alleviate colitis in mice by downregulating the expression of pro-inflammatory cytokine genes (IL-1β, IL-6, and TNF-α) and by upregulating the expression of tight junction protein genes (occludin and ZO-1).16Similarly, the intestinal protective effect of betaine has also been confirmed in different nutritional stress models in aquatic animals. Supplementing betaine in a low methionine diet can ameliorate the histopathological impairment of the intestines in grass carp(Ctenopharyngodon idella), upregulate the expression of tight junction protein genes in the fish intestines to maintain the intestinal barrier, and enhance antioxidant capacity.17 Supplementing betaine in the high-fat diets upregulates the mRNA expression levels of anti-inflammatory cytokines in the intestines of Black Seabream (Acanthopagrus schlegelii), while the expression levels of nuclear factor kB (NF-κB) and pro-inflammatory cytokines are downregulated, alleviating intestinal inflammation.18 The aforementioned studies are primarily based on terrestrial animals, herbivorous fish, and omnivorous fish. It remains unclear whether betaine can alleviate intestinal damage induced by the HC diets in carnivorous fish. Therefore, whether betaine can mitigate intestinal damage induced by the HC diets warrants further investigation.
The largemouth bass (also known as the California bass), is a species native to California, USA. Due to its advantages such as being rich in high-quality protein and nutrients and having tender and firm flesh, it has gradually become one of the popular seafood choices in recent years.19,20 Largemouth bass is a typical carnivorous fish characterized by a relatively short digestive tract, low activity of carbohydrate-digesting enzymes, and limited metabolic capacity for dietary carbohydrates, making it an ideal model organism for investigating carbohydrate intolerance in carnivorous fish. When the carbohydrate content in feed exceeds 15%, it leads to reduced antioxidant capacity, intestinal microbiota disorder, and upregulation of inflammation-related genes (IL-8, IL-1β, TNFα, and COX-2) in the intestine.21,22 Despite current advances in understanding the detrimental effects of the HC diets on fish intestinal health, the underlying mechanisms linking carbohydrate overload to intestinal dysfunction remain incompletely elucidated. Therefore, this study focuses on largemouth bass to investigate the effects of supplementation of betaine to the HC diets on the intestinal health of juvenile largemouth bass and to explore the feasibility of betaine as an aquafeed additive, providing theoretical guidance and reference for the reasonable use of carbohydrates and cost reduction in aquaculture for healthy fish farming.
2. Materials and methods
2.1. Feed preparation
This study used fish meal, Soy protein isolate, and gluten meal as protein sources, and fish oil as a fat source. 0.5%, 1.0%, 1.5% and 2.0% betaine were supplemented as diets, a range defined from previously published studies in fish.18,23,24 Six feed formulations were designed in Table 1, including a low carbohydrate diet (8% corn starch) (LC), a high carbohydrate diet (20% corn starch) (HC), and high-carbohydrate feed supplemented with 0.5% betaine (HCB1), 1.0% betaine (HCB2), 1.5% betaine (HCB3), and 2.0% betaine (HCB4). To prepare the feed, fish meal was first crushed using a grinder (SGF130, Shanghai Tianfan Pharmaceutical Machinery Co., China), and all ingredients were passed through a 60-mesh sieve, accurately weighed, and thoroughly mixed in ascending order of quantity. After mixing, the ingredients were processed into pellets with a diameter of 5 mm using a pelletizer (CD4 × 1 TS, South China University of Technology, Guangzhou, China). The prepared feed pellets were dried in an oven at 55°C, and once dry, stored in a -20°C freezer for later use. The experimental diets were subjected to aquatic stability testing, and the water solubility loss rate of the feed was less than 2%.
2.2. Fish and feeding trial
Largemouth bass were purchased from a fish farm in Zhangzhou City, Fujian Province, China. Largemouth bass were temporarily housed in circular tanks (1200 L) at the Aquaculture Experimental Farm of Jimei University for a period of two weeks. During the temporary housing, the fish were fed commercial feed twice daily at 9:00 AM and 5:00 PM until apparent satiety, feeding for each tank was completed within 5 minutes, and waste was suctioned half an hour after feeding. Water was partially replaced once each afternoon, with about 30% of the water being exchanged. After the temporary housing period, the fish were fasted for 24 hours and then anesthetized using a solution containing 150 mg/L of eugenol, purchased from Heshenfu Technology Co., Ltd. (Guangzhou, China). A total of 270 healthy, disease-free, and uniform-sized juvenile largemouth bass (4.34±0.11 g) were randomly assigned to 18 experimental tanks (65 L) for the aquaculture trial, divided into 6 groups with three parallel tanks per group, 15 fish per tank. The initial stocking density was 1.00 kg/m3. The aquaculture trial was conducted in a manner similar to the temporary housing, utilizing a recirculating water system. Dissolved oxygen, water temperature, pH, ammonia nitrogen, and nitrite levels were measured daily. The conditions were maintained to ensure a dissolved oxygen concentration of≥7 mg/L, a water temperature of 25±2℃, a pH range of 6.7-7.2, an ammonia nitrogen concentration of <0.1 mg/L, and a nitrite concentration of <0.08 mg/L.
2.3. Sample collection
After 56 days of cultivation, the experimental largemouth bass were fasted for 24 hours. The fish were anesthetized using a solution containing 150 mg/L of eugenol. From each experimental tank, 11 fish were randomly selected to record their body weight, body length, liver weight, and abdominal fat weight. Blood was collected via the caudal vein, left at 4°C for 20 hours, and centrifuged at 4°C and 3000 r/min for 10 minutes to obtain serum. The serum was stored at -80°C for biochemical indicator analysis. The 11 fish above were dissected to collect intestinal tissues. Among these samples, 9 were cut into equal portions, placed into cryovials, and rapidly frozen in liquid nitrogen. Nine of these equal portions were used for enzyme activity testing, and the other nine were used for quantitative real-time PCR analysis and gut microbiota analysis. The tissues were stored at -80°C. Additionally, the remaining 2 samples were fixed in 4% paraformaldehyde solution for HE section preparation.
2.4. Growth performance
Weight growth rate (WGR, %) = (Wb - Wa) / Wa ×100;
Specific growth rate (SGR, %/d) = (ln Wb - ln Wa) / d ×100;
Feed conversion rate (FCR)= FI / (Wb-Wa);
Feed intake (FI,g)=F/N
Intraperitoneal fat ratio (IPF, %) =WF/ WH×100
Hepatosomatic index (HSI, %) WL/ WH×100
Survival Rate (SR, %)= [(N0 - N1)/N0] ×100
Where Wb is the average final weight (g); Wa is the average initial weight (g); d is the experimental period in days; FI is the average feed intake per fish (g); F is the total feed intake per tank (g); N is the number of fish per tank; WL is the wet weight of the liver of the experimental fish (g); WH is the wet weight of the experimental fish (g); WF is the weight of the abdominal fat of the experimental fish (g). N0 is the initial number of fish per tank; N1 is the number of dead fish per tank.
2.5. Analysis of intestinal digestion and antioxidant indicators
We weighed the intestinal tissue and mixed it with physiological saline at a ratio of 1:9. Then, we homogenized it in a tissue grinder for 45 seconds. The mixture was centrifuged at 3000 r/min for 10 minutes at 4°C, and the supernatant was collected for the detection of digestive and antioxidant indicators. Commercial kits were used for the indicator measurements, all of which were purchased from the Nanjing Jiancheng Institute, and the tests were conducted strictly according to the manufacturer’s instructions. Intestinal digestive enzymes: amylase (AMS, Cat. No.: C016-1-2), lipase (LPS, Cat. No.: A054-2-1), trypsin (TRS, Cat. No.: A080-2-2). Intestinal antioxidant indicators: total antioxidant capacity (T-AOC, Cat. No.: A015-2-1), catalase (CAT, Cat. No.: A007-1-1), malondialdehyde (MDA, Cat. No.: A003-1-2), superoxide dismutase (SOD, Cat. No.: A001-3-2), trace reduced glutathione (GSH, Cat. No.: A006-2-1).
2.6. Analysis of serum biochemical parameters
D-lactic acid (D-LA), diamine oxidase (DAO), and lipopolysaccharide (LPS) activities were determined using commercial assay kits. The D-LA kit (Cat. No.: A19-3-1) was purchased from Nanjing Jiancheng Bioengineering Institute, the DAO kit (Cat. No.: ADS-W-QT043) from Aidesen Biotechnology Co., Ltd., and the LPS (Cat. No.: MM-9000702) kit from Jiangsu Enzyme Immuno Co., Ltd. All kits were used strictly following the manufacturers’ instructions.
2.7. Intestinal morphology analysis
Intestinal morphology was evaluated using hematoxylin and eosin (H&E) staining of paraffin-embedded sections. Briefly, samples were fixed in 4% paraformaldehyde solution for 24 hours. Following fixation, the tissues were trimmed in a fume hood to expose the target area and placed into embedding molds with corresponding labels. The tissues were dehydrated sequentially through a graded series of alcohols: 75% ethanol for 4 hours, 85% ethanol for 2 hours, 90% ethanol for 2 hours, 95% ethanol for 1 hour, anhydrous ethanol I for 30 minutes, anhydrous ethanol II for 30 minutes; then treated with xylene-alcohol for 5–10 minutes, xylene I for 5–10 minutes, xylene II for 5–10 minutes, followed by paraffin wax at 65°C: paraffin I for 1 hour, paraffin II for 1 hour, paraffin III for 1 hour. The paraffin-embedded tissues were then placed into an embedding machine (Manufacturer: Wuhan Junjie Electronics Co., Ltd., Model: JB-P5) for embedding. Melted paraffin was first poured into the embedding mold, and before the paraffin solidified, the tissue was taken from the dehydration box, placed in the mold according to the embedding orientation, and labeled. The mold was cooled on a -20°C freezing platform (Manufacturer: Wuhan Junjie Electronics Co., Ltd., Model: JB-P5). After the paraffin solidified, the paraffin block was removed from the mold and trimmed. The trimmed paraffin block was sectioned using a paraffin microtome (Manufacturer: Shanghai Leica Instruments Co., Ltd., Model: RM2016) at a thickness of 4 μm. The sections were floated on a slide-warming device (Manufacturer: Zhejiang Jinhua Kedi Instrument Equipment Co., Ltd., Model: KD-P) with water at 40°C to flatten the tissue, then lifted onto slides and baked in a 60°C oven (Manufacturer: Tianjin Laibori Instrument Equipment Co., Ltd., Model: GFL-230). After water and paraffin baking, the slides were taken out and stored at room temperature for later use. Before use, the sections were observed and imaged using a bright-field microscope (Eclipse Ci-L, Nikon, Japan), and the WT1000GM system was used to measure the height and width of intestinal villi and the thickness of the muscular layer in the tissue sections.
2.8. Gut microbiota analysis
16S rDNA gene sequencing was used to analyze the Gut microbiota. Genomic DNA from intestinal samples was extracted using the MagPure Soil DNA LQ Kit (Magan) according to the manufacturer’s instructions. The concentration and purity of the DNA were assessed using NanoDrop 2000 (Thermo Fisher Scientific, USA) and agarose gel electrophoresis, and the extracted DNA was stored at -20℃. The extracted genomic DNA was used as a template for PCR amplification of bacterial 16S rRNA genes with barcode-labeled specific primers and Takara Ex Taq high-fidelity enzyme. Universal primers 343F (5’-TACGGRAGGCAGCAG-3’) and 798R (5’-AGGGTATCTAATCCT-3’), or 515F (5’-GTGCCAGCMGCCGCGG-3’) and 907R (5’-CCGTCAATTCMTTTRAGTTT-3’), were used to amplify the V3-V4 (or V4-V5) variable regions of the 16S rRNA gene for bacterial diversity analysis. The PCR amplification products were examined by agarose gel electrophoresis.
2.9. Quantitative real-time PCR analysis
Details of primers used in this study are shown in Table 2. Total RNA was extracted from intestinal tissues using the SteadyPure RNA Extraction Kit (Accurate Biotechnology (Hunan) Co., LTD, China, Nanjing), and cDNA was synthesized from the total RNA using the Evo M-MLV RT Mix Kit with gDNA Clean for qPCR Ver.2 (Accurate Biotechnology (Hunan) Co., LTD, China, Nanjing) for qPCR. qPCR detection was performed using the SYBR Green Premix Pro Taq HS qPCR Kit (Accurate Biotechnology (Hunan) Co., LTD, China, Nanjing). The reaction system was 20 μL, including 10 μL SYBR Green Pro Taq HS Premix, 0.4 μL forward primer, 0.4 μL reverse primer, 2 μL cDNA template, and 7.2 μL RNase Free Water. The qPCR amplification program consisted of an initial denaturation at 95°C for 30 s; during the cycling process, 45 cycles were performed at 95°C for 5 s and 60°C for 30 s. After amplification, the melting curve was analyzed on a LightCycler®480 II. Quantification was performed using the 2−ΔΔCT method, with β-actin as the internal reference gene.
2.10. Statistics analysis
Statistical analysis of the experimental data was performed using SPSS 25.0 software. One-way analysis of variance (ANOVA) was used to analyze the differences among all experimental groups. If differences were found, Duncan’s method was used for multiple comparisons, or an independent samples t-test was used to analyze the data. All experimental results are presented as mean ± standard deviation, with “∗” denoting statistical differences among the groups (∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.01). Means without a common superscript differ significantly (P < 0.05). Figures were produced using GraphPad Prism 10.6.
3. Results
3.1. Growth performance
After eight weeks of feeding juvenile largemouth bass, the effects of betaine on the growth performance of largemouth bass are presented in Table 3. Compared with the LC group, there were no significant differences in WGR, SGR, FCR, and HSI between the HC group (P > 0.05). Compared with the HC group, WGR and SGR were significantly higher, and FCR and HSI were significantly lower in the HCB1 group (P < 0.05). Furthermore, HSI was significantly reduced in the HCB groups relative to the HC group (P < 0.05). Furthermore, HSI was significantly reduced in the HCB groups relative to the HC group (P < 0.05). There was no significant difference in IPF between treatments (P > 0.05).
3.2. Intestinal digestive capacity
The effects of betaine on the intestinal digestive enzyme activities of juvenile largemouth bass are shown in Table 4. Among the intestinal digestive enzyme indicators, the activities of LPS and TRP showed no significant differences between the groups (P>0.05). Compared with the LC group, the AMS activity in the HC group tended to increase, but the difference was not statistically significant (P>0.05). Compared with the HC group, the AMS activity in the HCB1 group tended to increase, but the difference was not statistically significant (P>0.05).
3.3. Intestinal antioxidant capacity
The effects of betaine on the intestinal antioxidant parameters of juvenile largemouth bass are shown in Table 5. Compared with the LC group, the activities of T-AOC, SOD, and GSH in the HC group were significantly reduced (P<0.05); CAT in the HC group showed a decreasing trend, and MDA in the HC group showed an increasing trend, but neither was statistically significant (P>0.05). Compared with the HC group, T-AOC and SOD in the HCB groups were significantly increased (P<0.05), and GSH in the HCB2 group was significantly increased (P<0.05); compared with the HC group, CAT in the HCB group showed an increasing trend, while HCB1, HCB2, and HCB3 groups showed a decreasing trend, but none of these were statistically significant (P>0.05).
3.4. Intestine morphology and physical barrier
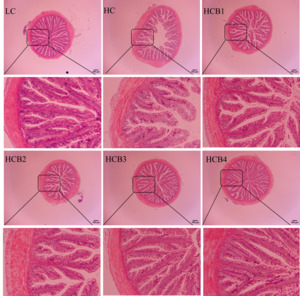
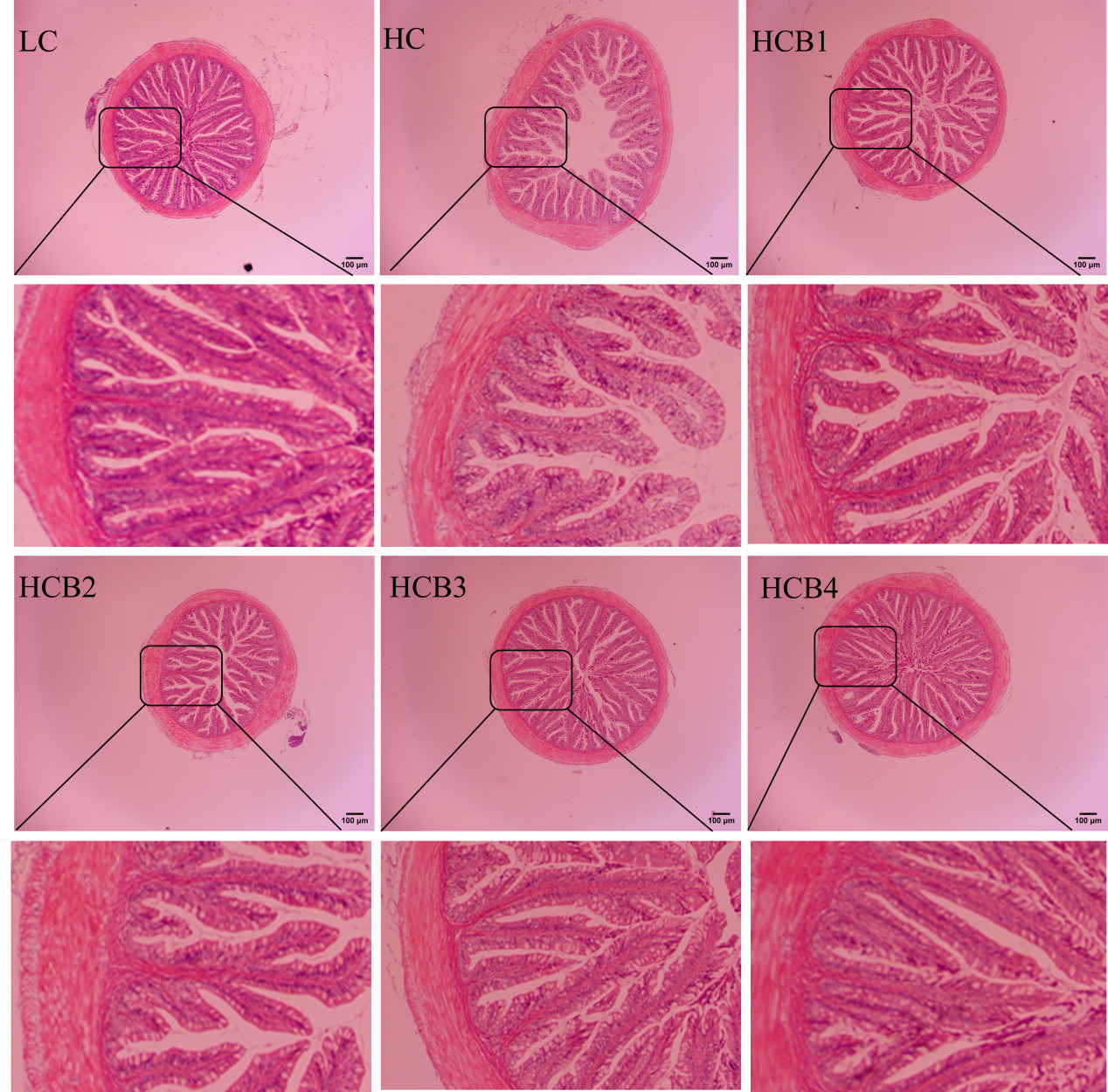
The intestinal morphology and structure of the largemouth bass are shown in Fig. 1 and Table 6. In the HC group, a distinct phenomenon of lamina propria detachment was observed. Compared with the LC group, the intestinal villus height and muscular thickness in the HC group were significantly decreased (P < 0.05); the intestinal villus thickness in the HC group showed a decreasing trend, but the difference was not statistically significant (P > 0.05). Compared with the HC group, the intestinal villus height in the HCB1, HCB3, and HCB4 groups was significantly increased (P < 0.05); the intestinal villus thickness in the HCB2 group was significantly increased; and the intestinal muscular thickness in the HCB2, HCB3, and HCB4 groups was significantly increased (P < 0.05).
The expression levels of intestinal tight junction (TJ) protein are shown in Fig. 2. Compared with the LC group, the expression levels of ZO-1, Occludin, and Claudin-1 in the HC group were significantly decreased (P<0.05). Compared with the HC group, the expression levels of ZO-1 and Occludin in the HCB groups were significantly increased (P<0.05), and the expression levels of Claudin-1 in HCB2 and HCB3 groups were significantly increased (P<0.05).
3.5. Permeability parameters of intestinal mucosal barrier
The permeability parameters of the intestinal mucosal barrier are shown in Table 7. Compared with the LC group, the DAO content was significantly increased in the LC group (P<0.05). Compared with the HC group, the DAO content in the HCB groups was significantly decreased (P<0.05), the DAO content was the lowest in the HCB1 group. No statistically significant differences in D-LA and LPS were detected among all groups (P> 0.05).
3.6 Gut microbiota
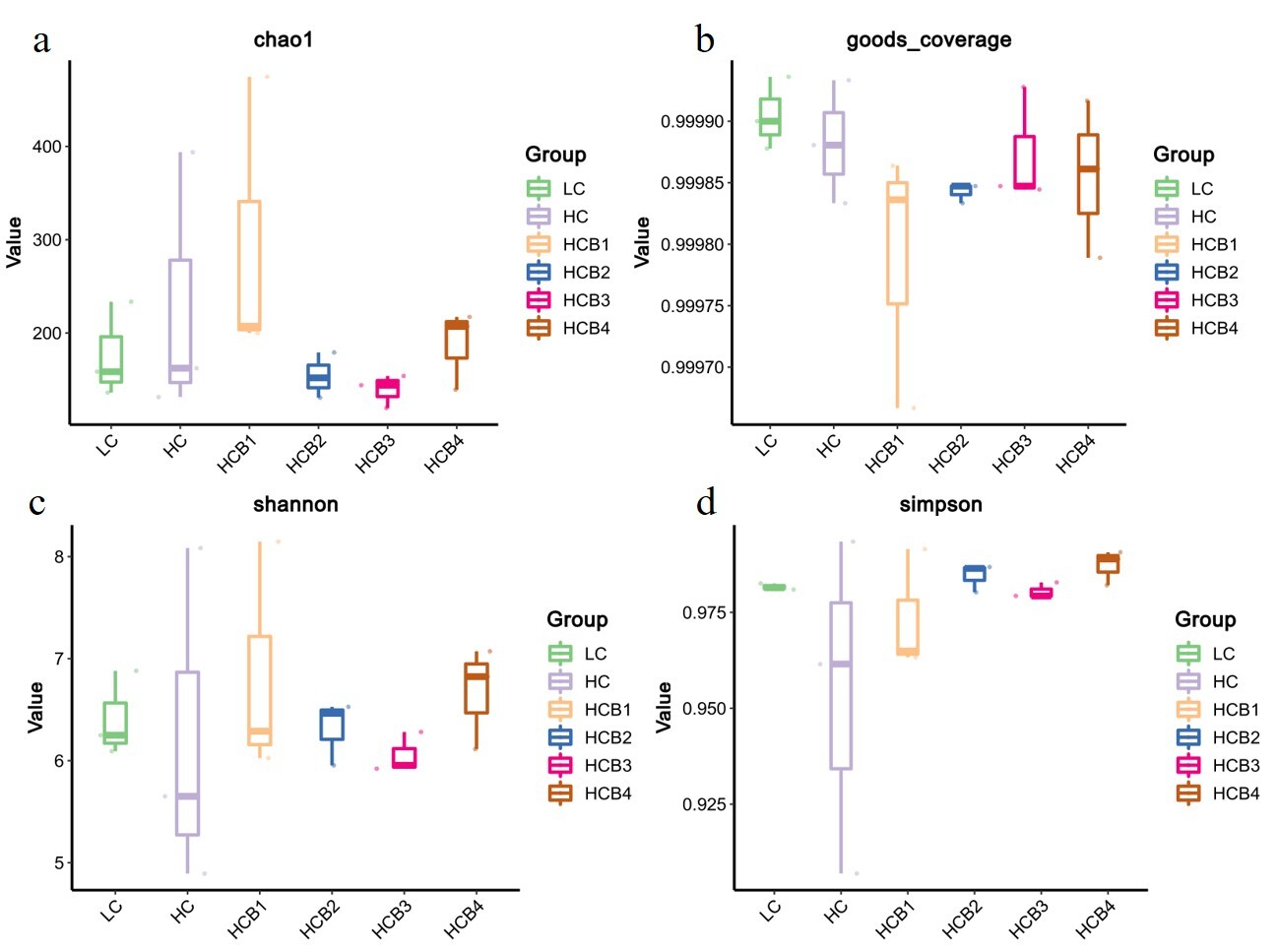
3.6.1. Alpha diversity analysis
The results of intestinal microbial alpha diversity are presented in Fig. 3. No significant statistical differences were detected in the Chao1, Goods_coverage, Shannon, and Simpson indices among all groups (P > 0.05).
3.6.2. Intestinal microbiota composition
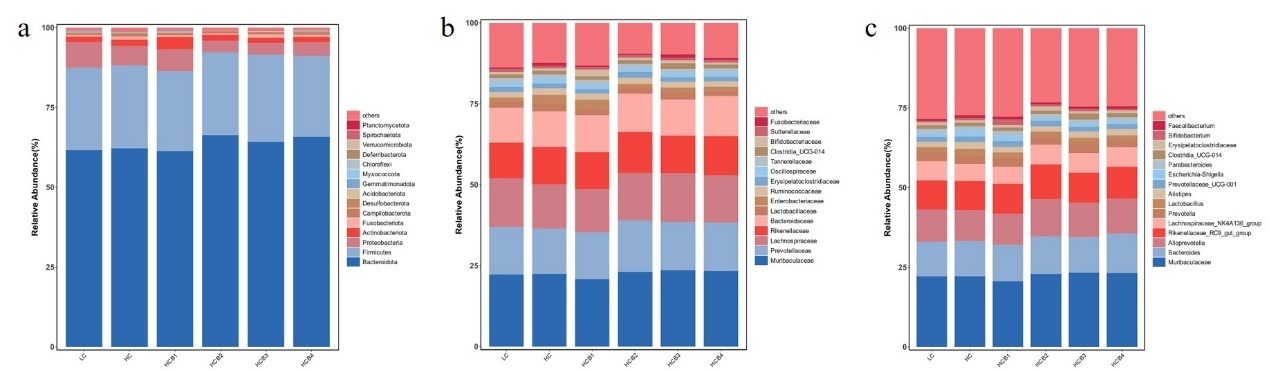
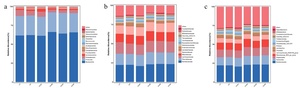
The relative abundances of the top fifteen bacteria at the phylum, family, and genus levels are shown in Fig. 4. The composition of the microbial community indicates that at the phylum level, Bacteroidota, Firmicutes, Proteobacteria, and Actinobacteriota dominate across all groups. Compared with the HC group, the relative abundance of Proteobacteria in the HCB2 and HCB3 groups was significantly decreased (P < 0.05), while the abundance of Actinobacteriota in the HCB1 group was significantly increased (P < 0.05). At the family level, Muribaculaceae, Prevotellaceae, Lachnospiraceae, and Rikenellaceae are dominant across all groups. Compared with the LC group, the abundance of Enterobacteriaceae in the HC group was significantly increased (P < 0.05), while compared with the HC group, the relative abundance of Enterobacteriaceae in the HCB2 and HCB3 groups was significantly decreased (P < 0.05). At the genus level, Muribaculaceae, Bacteroides, Alloprevotella, and Rikenellaceae_RC9_gut_group dominate across the groups studied.
3.7. Gene expression related to intestinal inflammation
The expression levels of intestinal inflammatory factors are shown in Fig. 5. Compared with the LC group, the HC group showed significantly increased expression levels of NF-κB, COX-2, IL-8, and IL-1β, while the expression levels of IL-10 and TGF-β1 were significantly decreased (P<0.05). Compared with the HC group, the relative expression levels of NF-κB, COX-2, and IL-8 were significantly decreased in the HCB1 and HCB2 groups; the expression level of IL-1β in the HCB groups showed a decreasing trend but was not statistically significant (P>0.05); the expression level of IL-10 in the HCB groups were significantly increased (P<0.05), reaching the maximum in the HCB1 group; the expression levels of TNF-β1 in HCB2 and HCB3 groups were significantly increased (P<0.05).
4. Discussion
4.1. Growth Performance
In this study, dietary starch levels did not significantly affect the growth performance of largemouth bass, which is consistent with previous research findings.25 In this study, supplementation of 0.5% betaine in the HC diets improved the growth performance of juvenile largemouth bass. In previous studies on Nile tilapia (Oreochromis niloticus), compared with diets containing 0%, 1%, 1.5%, 2%, and 2.5% betaine, fish fed with 0.5% betaine showed the best weight gain rate (WGR) and specific growth rate (SGR).26 Compared with the 1% and 1.5% betaine groups, supplementing 0.5% betaine led to the best growth performance of giant river prawn(Macrobrachium rosenbergii), which is consistent with the results of this study.27Similarly, supplementing 0.8% betaine in the HC diets resulted in the highest weight gain rate (WGR) and specific growth rate (SGR) and the lowest feed conversion ratio (FCR) in blunt snout bream(Megalobrama amblycephala).28 This may be due to differences in fish species and size, leading to different betaine requirements. In this study, betaine supplementation was able to reduce HSI, which is consistent with research on blunt snout bream, possibly due to betaine improving liver damage by ameliorating liver pathological lesions.29,30 The above results indicate that 0.5% betaine can promote the growth of largemouth bass.
4.2. Intestinal antioxidation
The antioxidant indicators in an animal’s body can directly or indirectly reflect its health status. T-AOC is an indicator that measures the total antioxidant capacity in an organism and evaluates the body’s redox capability.31 SOD and CAT are widely present antioxidant enzymes in organisms; SOD primarily serves to remove superoxide radicals within cells by catalyzing their conversion into hydrogen peroxide, while CAT mainly functions to eliminate hydrogen peroxide within cells.32,33GSH is an important non-enzymatic antioxidant in the body.34 MDA is a major product of lipid peroxidation and is also a biomarker of oxidative stress.35 In this study, the HC diets reduced intestinal T-AOC, SOD, and GSH activity, which returned to relatively normal levels after the addition of betaine. Previous studies have shown that long-term carbohydrate intake reduces the intestinal antioxidant capacity of largemouth bass, consistent with the results of this study.22Betaine is widely used as an antioxidant in poultry, but research in aquaculture is limited.36 In studies on zebrafish, dietary betaine concentrations of 0.1, 0.2, and 0.4 g/kg enhanced the gene expression of superoxide dismutase (SOD), glutathione peroxidase (GSH-PX), and catalase (CAT) in the liver.37 Supplying 1% betaine to the HC diets alleviates oxidative stress induced by regulating the expression of antioxidant enzymes.38 Mechanistically, betaine converts homocysteine into methionine, which is subsequently transformed into S-adenosylmethionine (SAM), and SAM participates in GSH metabolism.39 In this study, betaine was able to increase T-AOC, SOD, and GSH levels in the intestine of largemouth bass, possibly related to betaine’s involvement in GSH metabolism, which contributes to enhancing cellular antioxidant defense mechanisms and protecting cells from oxidative damage.40 These results indicate that betaine can mitigate high-carbohydrate diet-induced oxidative stress and improve the intestinal antioxidant capacity of largemouth bass.
4.3. Intestinal Structure and Intestinal Mucosal Permeability
The villus height, villus thickness, and muscular thickness of fish intestines are important histological indicators for evaluating the intestinal health status of fish.41 In this study, the HC diets reduced villus height and muscular thickness, but betaine reversed these effects. Previous studies have shown that HC diets damage the intestinal tissue morphology of blunt snout bream, snakeheads (Channa argus), and largemouth bass, which is consistent with the results of this study.42,43Supplementation with betaine in the HC diets increased intestinal villus length, which aligns with previous research in Nile tilapia.44These results may be due to the accumulation of betaine, enhancing the water-holding capacity of intestinal cells, thereby restoring the structure of intestinal epithelial cells.45,46 It is well-known that the integrity of the intestinal structure is related to tight junction proteins in the intestine. The tight junction (TJ) proteins include transmembrane proteins (the Claudin family and Occludin) and cytoskeleton-associated proteins (zonula occludens (ZO) proteins).47In this study, we found that the HC diets significantly downregulated the expression levels of ZO-1, Occludin, and Claudin-1, consistent with previous studies in golden pompano (Trachinotus ovatus).48However, we demonstrated that betaine could improve the expression of the TJ protein gene in HC-induced juvenile largemouth bass, further indicating the protective effect of betaine on the intestinal barrier. Furthermore, in previous studies with indigenous broilers, supplementation with betaine (1000 mg/kg) improved the expression of the TJ protein in the small intestine of native broilers exposed to high-temperature environments.49 In earlier studies with pigs, maternal betaine supplementation improved the expression of intestinal nutrient transporter genes in weaned piglets.50This is similar to the results of this study, but reports on the protective effects of dietary betaine against downregulation of intestinal the TJ or AJ proteins in fish are very limited, and the specific mechanisms still need further investigation. In addition, reduced expression or structural damage of Occludin, Claudin-1, and ZO-1 can lead to dysfunction of intestinal epithelial cells, resulting in increased intestinal mucosal permeability.51Serum D-LA and DAO are markers reflecting intestinal mucosal damage and intestinal permeability.52,53Only small amounts of DAO and D-LA exist in the animal circulatory system, but when the intestinal mucosal barrier is damaged, serum levels of D-LA and DAO increase, indicating enhanced intestinal mucosal permeability.54 In this study, HC increased serum DAO levels, while betaine supplementation reversed this effect, possibly due to betaine’s ability to regulate tight junction protein genes. The above results indicate that betaine can reduce intestinal mucosal permeability and improve intestinal structure.
4.4. Gut microbiota
The gut microbiota forms a microbial barrier in the intestine and plays a crucial role in maintaining intestinal integrity.55 In the present study, the dominant bacterial phyla in the intestine of largemouth bass were Bacteroidota, Firmicutes, Actinobacteriota, and Proteobacteria, which is consistent with the findings of previous studies.56At the phylum, family, and genus levels, there were no significant differences in the dominant microbiota between the LC and HC groups. This observation is similar to the results reported in Atlantic salmon (Salmo salar),57 possibly because an increase in dietary digestible carbohydrate levels primarily affects low-abundance bacteria in the intestine. Notably, although Enterobacteriaceae was not a dominant family in the gut, its relative abundance was significantly increased in the HC group. Previous studies have demonstrated that Enterobacteriaceae contain molecules that directly enhance inflammatory responses, which can induce the secretion of inflammatory factors such as IL-8 and IL-1β, disrupt intestinal mucosal tight junctions, and increase intestinal permeability.58 These results suggest that a high-carbohydrate diet induces intestinal inflammation in largemouth bass, while supplementation with 1.0% and 1.5% betaine in the HC diets significantly reduced the relative abundance of Enterobacteriaceae. In the HC diets,1.0% and 1.5% betaine significantly decreased the relative abundance of Proteobacteria, whereas 0.5% betaine significantly increased the relative abundance of Actinobacteriota. Previous studies have confirmed that most bacteria in Proteobacteria are pathogenic; their overproliferation can disrupt the balance of the gut microbiota and induce intestinal inflammation.59 In contrast, Actinobacteria can synthesize various bioactive products that exert beneficial effects on fish health, including enhancing antibacterial activity against pathogens, regulating immune responses, and promoting growth.60 Based on these findings, we speculate that the optimal growth performance observed in the group supplemented with 0.5% betaine in the HC diets may be closely associated with the increased relative abundance of Actinobacteria. Alpha-diversity indices (chao1, goods_coverage, Shannon and Simpson) did not differ among treatments, indicating that betaine did not alter the diversity of the gut microbiota but significantly improved the intestinal microbial composition. A previous study on broilers reported that betaine maintains the intestinal microbial barrier by increasing the abundance of beneficial bacteria and decreasing the abundance of harmful bacteria,61 which is consistent with the regulatory effect of betaine on the gut microbiota of largemouth bass in the present study. This regulatory mechanism may be attributed to the fact that betaine can provide additional carbon and nitrogen sources for intestinal symbiotic bacteria,62 thereby optimizing the microbial composition. It should be noted that the differences in feed composition between the LC and HC groups included not only an increase in digestible carbohydrates (corn starch) but also a decrease in microcrystalline cellulose. However, the potential effects of cellulose on the gut microbiota and host metabolism in fish remain unclear, which warrants further investigation in future studies.
4.5. Inflammatory Response
The expression of intestinal immune regulatory molecules (cytokines) is crucial for the immunity of fish organisms.63 Intestinal inflammatory responses are closely related to intestinal health and are usually assessed by the expression levels of inflammatory cytokines.64Fish cytokines can be divided into anti-inflammatory factors (such as IL-10 and TGF-β1) and pro-inflammatory factors (such as IL-1β, COX-2, and IL-8).65The transcription factor nuclear factor-kappa B (NF-κB) is an important upstream signaling factor. When NF-κB is activated, it can translocate to cells to regulate inflammatory factors, promoting the expression of pro-inflammatory factors (such as IL-1β, COX-2, and IL-8).66 In this study, the HC diets activated the expression of NF-κB, IL-1β, COX-2, and IL-8, while inhibiting the expression of IL-10 and TGF-β1. This may be related to the increased relative abundance of Enterobacteriaceae mentioned above, as Enterobacteriaceae contain molecules that directly enhance inflammatory responses and can induce the secretion of IL-8, TNF-α, and IL-1β. These results indicate that the HC diets induce intestinal inflammation in juvenile largemouth bass, consistent with previous studies on largemouth bass.7 However, supplementation with 0.5%-1% betaine suppressed the expression of NF-κB, COX-2, and IL-8, while activating the expression of IL-10. It is well-known that betaine possesses anti-inflammatory effects in various diseases. In a previous study on rats, supplementation with betaine in streptozotocin-induced diabetic rats reduced blood markers of inflammation, namely TNF-α and IL-1β.67Moreover, in studies on black seabream, 2% betaine to a high-Fat-Diet significantly upregulated the intestinal expression of NF-κB, TNF-α, and TNF-α, and significantly upregulated the expression of il-10 in the intestine, which is similar to the results of this study.18 The supplementation level of betaine in black seabream is higher than that in largemouth bass, which may be attributed to the fact that black seabream has a thicker intestinal mucosa, requiring a higher concentration of betaine for intestinal barrier repair. Additionally, as a marine fish, black seabream has a higher demand for osmolytes than freshwater fish, and betaine, acting as an osmolyte, further increases the required betaine supplementation level. This may be due to betaine’s ability to inhibit NF-κB signaling, exerting anti-inflammatory effects. Mechanistically, betaine acts by maintaining thiol levels (especially GSH) to suppress ROS generation and NF-κB activity.68Additionally, the above data suggest that the addition of betaine to the HC diets lead to downregulation of pro-inflammatory factors and upregulation of anti-inflammatory factors, indicating that betaine may alleviate intestinal inflammatory responses by regulating the balance between pro-inflammatory and anti-inflammatory genes.
5. Conclusion
This study demonstrated that supplementation with 0.5% betaine improved the growth performance of juvenile largemouth bass. Betaine also enhanced antioxidant capacity, with the 1% supplementation level significantly increasing glutathione (GSH) levels. Furthermore, betaine supplementation at 0.5-1% downregulated the expression of genes mediated by the nuclear factor kappa-B (NF-κB) signaling pathway, while simultaneously upregulating the expression of anti-inflammatory genes and downregulating the expression of pro-inflammatory cytokines. Notably, betaine significantly strengthened both the intestinal mucosal barrier and physical barrier; specifically, the 0.5% betaine group exhibited the lowest diamine oxidase (DAO) levels. Additionally, betaine supplementation ranging from 0.5% to 1.5% exerted a positive influence on the intestinal microbial composition. Overall, betaine alleviates high-carbohydrate -induced intestinal injury by enhancing anti-inflammatory properties and strengthening intestinal barrier function. However, it is necessary to establish an intestinal injury model in subsequent experiments to gain a more comprehensive understanding of betaine’s effects on intestinal injury in juvenile largemouth bass. Furthermore, appropriate models should be developed to investigate the species-specific effects of betaine, evaluate its potential adverse impacts, and determine its optimal supplementation level.
Acknowledgments
This research was supported by the Science and Technology Program of Fujian Province (Grant No. 2015N0010) and the Science and Technology Program of Xiamen City (Grant No. 3502Z20143017).
Authors’ Contribution
Conceptualization: Wanting Luo (Equal), Zhongbao Li (Equal). Methodology: Wanting Luo (Lead), Zhangfan Huang (Equal), Yanbo Zhao (Equal), Longhui Liu (Equal). Formal Analysis: Wanting Luo (Lead). Writing – original draft: Wanting Luo (Lead). Writing – review & editing: Wanting Luo (Lead). Resources: Zhongbao Li (Lead). Funding acquisition: Zhongbao Li (Lead).
Competing Interest – COPE
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Conduct Approval – IACUC
All contents of this study are subject to the Regulations on the Administration of Experimental Animals (amended on March 1, 2017 by the National Animal Control Regulations of the People’s Republic of China No. 11 and Laws and Regulations of The State Council No. 676), and approved by the Animal Experiment Ethics Committee of Jimei University (Xiamen, China), approval code: JMU202103009.
Informed Consent Statement
All authors and institutions have confirmed this manuscript for publication.
Data Availability Statement
All are available upon reasonable request.

_protein_of.svg)

_protein_of.svg)