Introduction
White spot disease caused by Cryptocaryon irritans is one of the most common and economically devastating parasitic diseases in marine aquaculture, particularly in marine cage-culture and recirculating aquaculture systems.1–4 The causative agent, C. irritans, is a ciliate protozoan, parasitizing the skin, gills, and fins of marine fish.1,5,6 The infected fish exhibit reduction of respiratory, appearance of small white spots, reduced feeding, and witnessed a huge mortality if not promptly diagnosed and treated.1,7–9 In Vietnam, the rapid expansion of marine fish farming, particularly of high-value species such as golden pompano (Trachinotus blochii), red snappers (Lutjanus spp.), and groupers (Epinephelus spp.), has significantly contributed to the coastal economy.10,11 However, this intensification has also heightened the risk of parasitic disease outbreaks, including those caused by C. irritans.
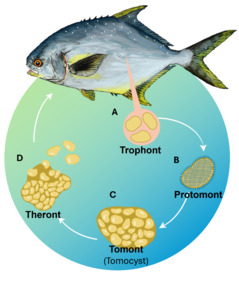
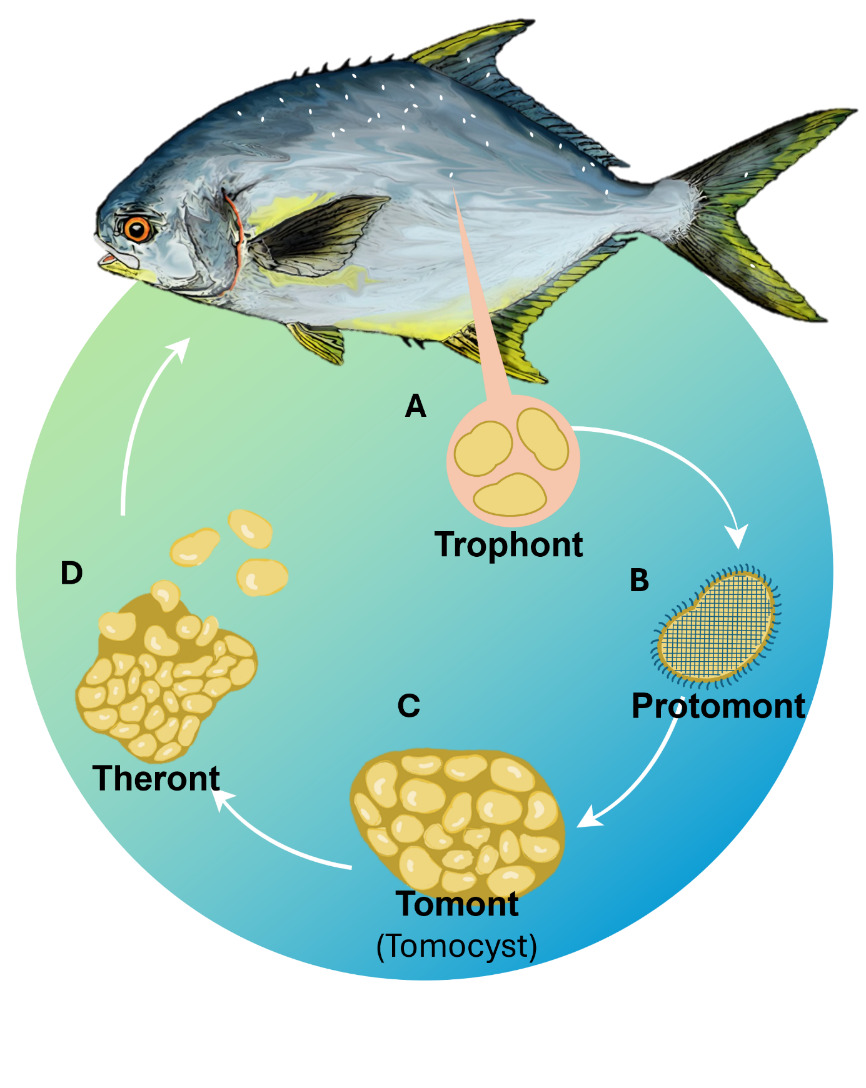
Due to its complex life cycle (Fig. 1), prolonged environmental persistence ability, and resistance to unfavourable conditions, most chemical treatments are only effective against the free-swimming theront stage. The tomont, in contrast, can survive in the environment for several days and exhibits resistance to common disinfectants due to the formation of thick-walled cysts. These features make the effective treatment of C. irritans in aquaculture systems particularly challenging.1,3,12–14
By the increasing prevalence of white spot disease in marine fish cultured and the growing demand for a treatments compound with low residue and high selectivity, Ivermectin has emerged as a potential candidate. Ivermectin - a semi-synthetic derivative of avermectin, isolated from the fermentation of Streptomyces avermitilis.15 It has demonstrated strong efficacy and has been widely used in veterinary medicine since the 1980s for treating nematode infections, later extending to human medicine especially for filariasis with long-lasting effects.16–19 Its primary mechanism of action involves enhancing gamma-aminobutyric acid (GABA) neurotransmission at neuromuscular junctions, leading to parasite paralysis and subsequent expulsion from the host.20
In aquaculture, Ivermectin has previously been studied for the treatment of monogenean infections21–23; digenean trematodes such as Dolfustrema bagarii24,25; myxozoan parasites like Myxobolus sp. and Thelohanellus kitauei.26,27 However, there is no published studies have evaluated the survival and developmental capacity of C. irritans when exposed to Ivermectin under in vitro conditions. Accordingly, this study was conducted to assess the mortality and development ability of C. irritans at the tomont and theront stages following exposure to varying concentrations of Ivermectin, providing a basis for future therapeutic trials and practical applications in the control methods of parasitic diseases in marine aquaculture.
Materials and Methods
Study duration and location
This study was conducted from May to June 2025 at the Laboratory of Aquatic Environment and Fish Pathology, Faculty of Fisheries, Vietnam National University of Agriculture. The Cryptocaryon irritans parasites used in this research were isolated from naturally infected golden pompano (Trachinotus spp.) collected in Quang Ninh Province, Vietnam. The isolates were subsequently maintained and propagated under laboratory conditions following the protocol of Watanabe, How,28 with a modifications adapted to local conditions in Vietnam.
The given Ivermectin’s low water solubility (1% injectable solution, VIC Animal Health – Russia), the commercial injectable formulation (solution in propylene glycol and glycerol formal) was diluted directly into sterile seawater (30‰ salinity). No additional solvents or surfactants were used. The control containing equivalent diluent (without ivermectin) was included to ensure the observed effects were due solely to ivermectin.
All experiments were conducted in Falcon 24-Well plates (2 cm diameter wells) under controlled temperatures ranging from 28-30℃.
Collecting C. irritans for experiment
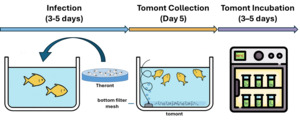
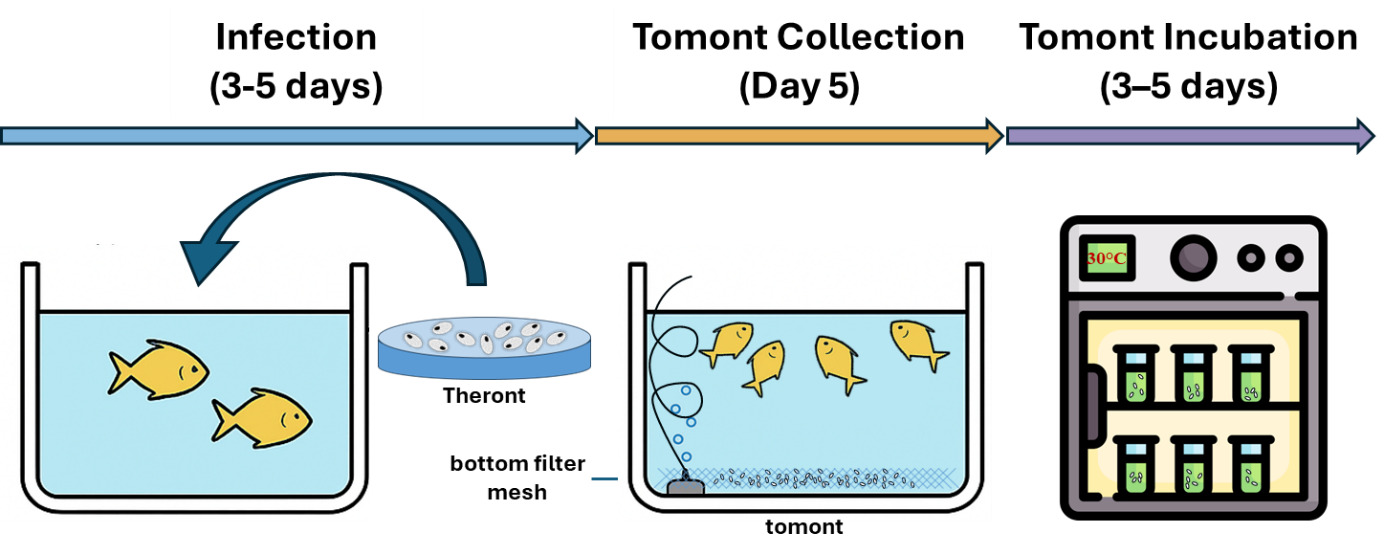
The C. irritans used in the experiment were sourced from laboratory-propagated parasites derived from golden pompano, following the methodology of,28 are simulated as shown in Fig. 2. Immediately, after trophonts exited the host, tomonts were collected using a bottom filter mesh system and filtered (30-50 µm). Samples were gently centrifuged at 1,500 rpm for 3 minutes. The pellet containing trophonts was retained and incubated in sterile seawater (30‰) at 28-30℃ to allow progression to the protomont stage.
After 18-24 hours, the protomonts (the stage that trophonts transitioning to tomonts) were examined microscopically to exclude deformity individuals. Selected protomonts (10 per well) were transferred into Falcon 24-Well plates (1 mL of sterile seawater per well). Each well was considered an independent experimental unit. Theronts hatched from these tomonts were subsequently used in the second experiment.
Experimental design
Two independent experiments were conducted to evaluate the effects of Ivermectin on two critical life stages of C. irritans: (1) the transformation and development of tomonts, and (2) the survival of theronts. Chemical exposure experiments were carried out with modifications based on Watanabe, Hansen.29 The concentration range (0.05–1.00 ppm) was selected based on previous aquatic toxicology and antiparasitic studies reporting safe and sublethal levels for fish.21,24,30 Concentrations below 1.0 ppm were considered within environmentally acceptable thresholds for short-term exposure.
Experiment 1: Effect of Ivermectin on the tomont development
This experiment assessed the impact of Ivermectin on the development of protomonts into mature tomonts and their ability to release theronts. Each well of a 24-Well plate received 10 protomonts in 1 mL of sterile seawater (30‰), with Ivermectin at concentrations of 0.05, 0.08, 0.10, 0.12, 0.16, 0.20, 0.50, and 1.00 ppm, along with a control group (0 ppm, sterile seawater only). Each treatment was performed in triplicate (three independent wells).
The plates were incubated at 28-30℃ in a temperature-controlled environment. The development of protomonts into tomonts, cyst formation, and theront hatching were monitored at 24 and 48 hours. The development of tomonts was evaluated by monitoring their proliferation under a light microscope.
Experiment 2: Effect of Ivermectin on theront survival
This experiment examined the direct impact of Ivermectin on the survival and motility of theronts – the infective stage of C. irritans. Theronts were collected immediately after being released from tomonts under artificial conditions and their initial density estimated using a Neubauer haemocytometer. Each well was loaded with 20-25 theronts in 1 mL of sterile seawater containing Ivermectin at the same concentrations as in Experiment 1 (0.05-1.00 ppm), with three replicates per concentration. The experiment was conducted at 28℃, and theront mortality was assessed after 60 minutes of exposure. Theronts were considered alive if they showed active swimming and retained a normal morphology. Non-motile or morphologically deformed theronts were recorded as dead.
Data analyse
All data were compiled and statistically analyzed using SPSS version 27.0 (IBM Corp., USA). Prior to conducting the one-way analysis of variance (ANOVA), the assumptions of normality and homogeneity of variances were assessed using the Shapiro-Wilk and Levene’s tests, respectively. A one-way ANOVA was then performed to evaluate significant differences among treatments, with the level of significance set at p < 0.05. When significant effects were detected, the Least Significant Difference (LSD) post hoc test was conducted to determine pairwise differences between treatment groups. Each treatment was replicated three times (n = 3) and arranged in a completely randomized design. Results are presented as mean values ± standard error (SE).
Results
Effects of Ivermectin on the development of trophonts
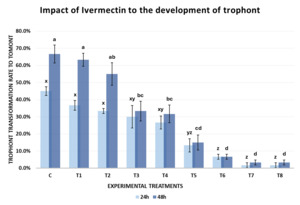
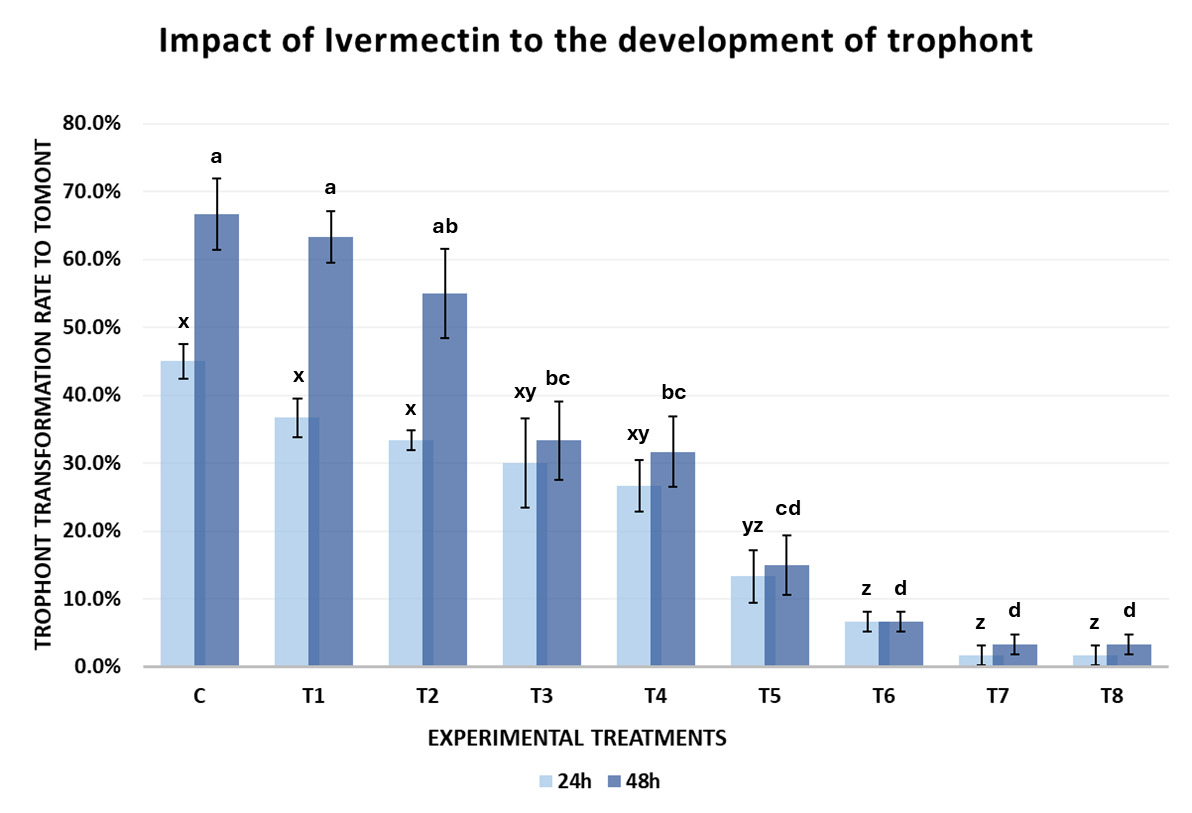
The results of Experiment 1 demonstrated that Ivermectin influenced the development of C. irritans from the protomont to fully formed tomont stage under in vitro conditions. The tomont transformation rate after 24 and 48 hours declined progressively with increasing concentrations of Ivermectin (Fig. 3).
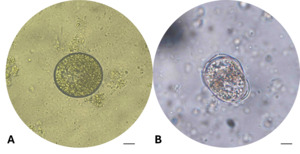
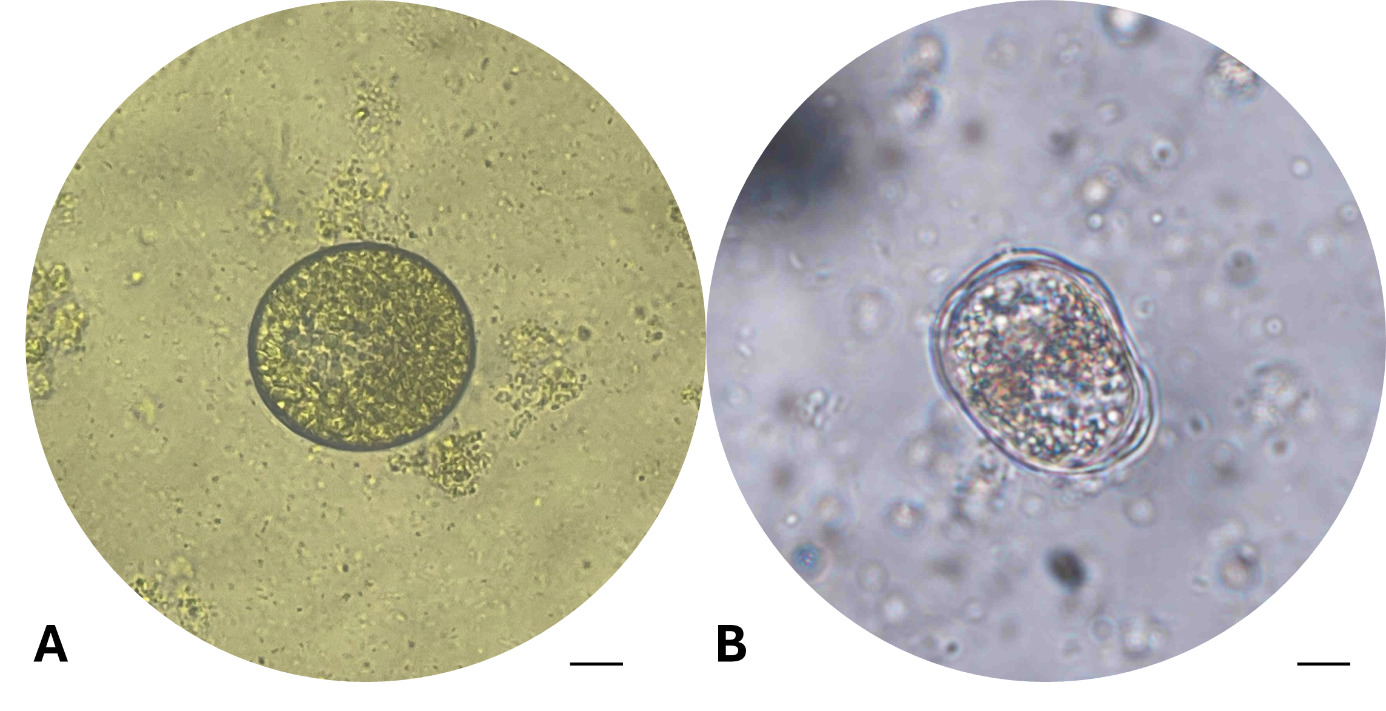
After 24 hours of incubation at 28-30℃, the tomont transformation rate in the control group (C-0 ppm) reached 45.00±5.00%, tomonts developed normally into rounded tomonts, thick and well-defined cyst wall, and dense (Fig. 4-A). In the low concentration treatments group (T1-T4; 0.05-0.12 ppm), the transformation rate remained relatively high (26.67-36.67%), and there were no statistically significant differences compared to the control (One-way ANOVA, p > 0.05). However, from the concentration of 0.16 ppm (T5) upwards, tomont development decreased markedly (1.67-13.33%), with statistically significant differences observed (p = 0.042–0.004 < 0.05), the tomonts exposed at 0.16 ppm Ivermectin exhibited abnormal morphology, including thinner cyst walls (Fig. 4-B). Specifically, at 0.20 ppm (T6), the tomont development rate dropped to 6.7%, and at the highest concentrations of 0.5-1.0 ppm (T7 and T8), the transformation rate was as low as 1.67±2.89%, indicating a strong inhibitory effect of Ivermectin on tomont development.
After 48 hours, the inhibitory trend became clearer. The control group (C) exhibited the highest tomont transformation rate at 66.67±10.41%. Meanwhile, groups T1 and T2 (0.05-0.08 ppm) still maintained high transformation rates (≥ 55%) without significant differences from the control (p > 0.05). From 0.10 ppm (T3) onwards, a notable decline in tomont development was observed (p = 0.039). At 0.16 ppm (T5), only 15.00±8.66% of tomonts developed successfully (p = 0.011). Particularly, at 0.5 and 1.0 ppm (T7 and T8), the development rate declined to 3.33±2.89%, nearly 20 times lower than the control, with statistically significant differences (p < 0.001).
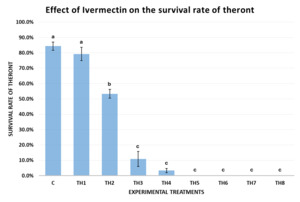
Effects of Ivermectin on the survival of theronts
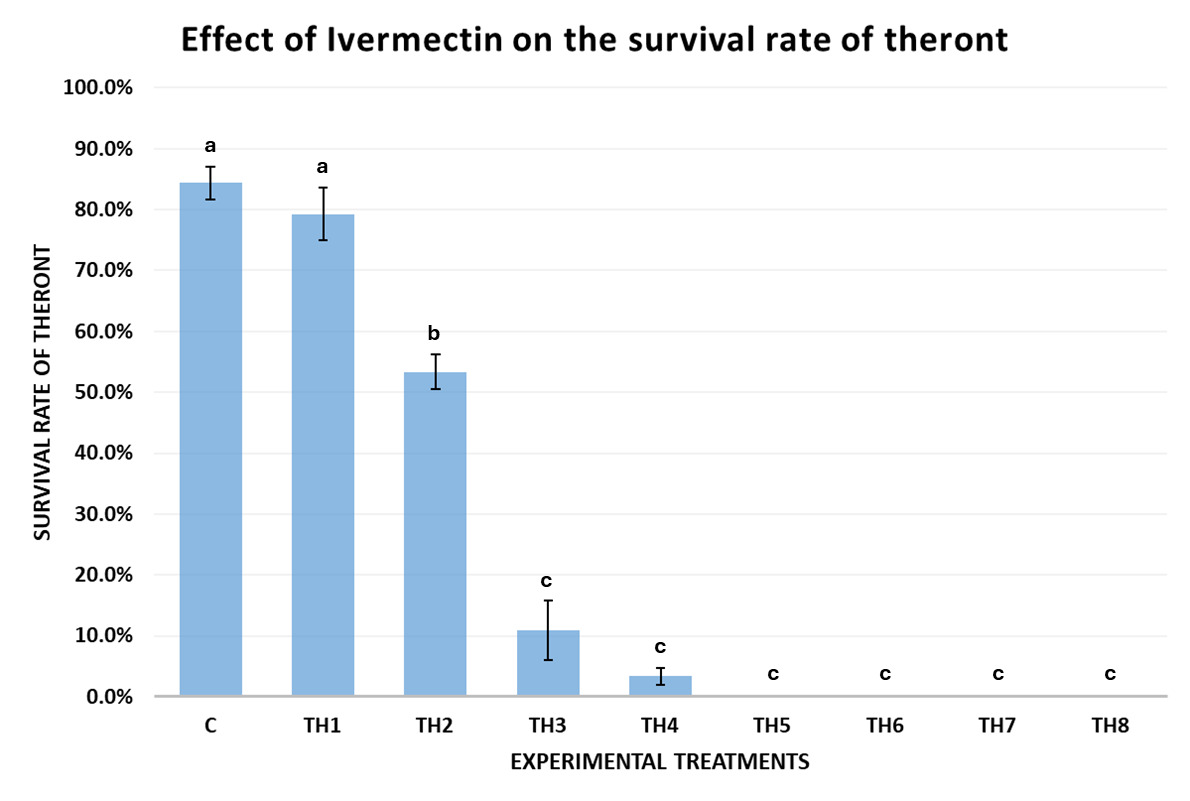
The results of Experiment 2 (Fig. 5) demonstrated that Ivermectin affected the survival rate of theronts after 60 minutes of exposure. In the control group (0 ppm), the theront survival rate reached 84.39±5.43%, indicating that experimental conditions did not affect their natural viability. When exposed to a low concentration of Ivermectin (TH1 – 0.05 ppm), the survival rate remained high (79.25±8.69%), with no statistically significant difference from the control (p = 0.24 > 0.05).
A significant reduction in survival was first recorded at 0.08 ppm (TH2), where the theront survival rate dropped to 53.33±5.77% after 1 hour, showing a statistically significant difference from the control (p < 0.001). Theront motility declined gradually from 0.10 ppm onwards, showed the strong effect of Ivermectin. At 0.10 ppm (TH3), the survival rate dropped to 10.89±9.82%, which was lower than the control (p < 0.0001) and TH1 (p = 0.007). continuing to decrease at higher concentrations. At 0.12 ppm (TH4), the survival rate further declined to 3.33±2.89% (p < 0.0001).
Notably, at concentrations of 0.16 ppm and above (0.16, 0.20, 0.50, and 1.00 ppm), all theronts became deformed or dissolved into the solution after 60 minutes of exposure, with none observable under the microscope, resulting in a presumed 100% mortality rate with statistically significant differences between treatments (p < 0.0001).
Discussion
Cryptocaryon irritans possess complex life cycles comprising three distinct stages: the free-swimming theront, the parasitic trophont, and the encysted tomont. Each stage necessitates targeted treatment strategies. Ivermectin has not traditionally been prioritised for use against ciliates, likely due to the biological differences between these protozoan parasites and the helminths or arthropods against which Ivermectin is typically effective. Concerns regarding its toxicity to aquatic organisms and its potential environmental persistence have further limited its application in aquaculture settings.30 Existing research has primarily focused on Ivermectin’s efficacy against nematodes such as Ascaris lumbricoides, Strongyloides stercoralis, Onchocerca volvulus, Wuchereria bancrofti; ectoparasites like Sarcoptes scabiei31 and Pediculus humanus32,33; digenean trematodes as Dolfustrema bagarii24,34; or crustacean parasites such as Lepeophtheirus salmonis35,36 and Lernaea spp.37 While some reports have indicated its efficacy against ectoparasitic insects and larval nematodes, Ivermectin appears to have limited evidence against protozoan, suggesting that its potential impact on ciliates such as C. irritans need further investigation.
With the other chemical agents, several studies have evaluated the inhibition of C. irritans tomonts under in vitro conditions. Zhan, Miao38 reported that copper sulphate inhibited tomont development at 15 ppm following a 3-hour exposure. However, nano-copper demonstrated minimal efficacy, likely due to structural limitations.39 The biosurfactant H6 from Pseudomonas exhibited inhibitory activity at 100 µg/mL,40 while Biokos – a polypeptide extract from Pseudomonas – achieved significant tomont and theront mortality within 1 hour at 48 mg/L,29 and 100% theront mortality at just 10 µg/mL after 30 minutes of exposure.40 Leptomycin B has also demonstrated broad-spectrum antiparasitic effects on both tomonts and theronts of C. irritans.41
In the present study, ivermectin demonstrated a markedly lower effective concentration (≥0.16 ppm), suggesting strong potency at sub-ppm levels. However, several other compounds achieved more rapid theront mortality, indicating that ivermectin may act more slowly but with a longer residual effect. For instance, honokiol extract42 from Magnolia bark inhibited tomont formation at 1.00 µg/mL after 8 hours and induced complete theront mortality at the same concentration within 30 minutes, while hydroxyl radical solution treatments43 at 0.5 mg/L inactivated theronts within 2 minutes. These comparisons highlight ivermectin’s potential advantage in sustained control, although its environmental persistence warrants further investigation.
The molecular mechanisms underlying ivermectin’s antiparasitic effects on ciliates remain unclear. Ivermectin primarily targets glutamate- and GABA-gated chloride channels in invertebrates.20 While genomic data for C. irritans are limited, the presence of GABA-like receptors in related ciliates such as Paramecium primaurelia suggests possible neuromodulatory effects.44 Alternatively, the observed deformities and rapid mortality may indicate nonspecific membrane disruption or interference with ion transport, as previously observed in protozoan models.
However, further studies are required to assess potential toxicity to cultured marine fish, bioaccumulation in tissues, environmental effect, and the feasibility of practical application. Future research could explore field-based treatments, including bath administration in juvenile fish, oral delivery, or injectable routes, possibly in conjunction with hepatoprotective agents, detoxifiers, and boosting immune stimulants to optimise therapeutic efficacy and reduce disease prevalence in marine aquaculture.
Conclusion
This study is among the first in Vietnam to evaluate the effects of Ivermectin on the parasite C. irritans under in vitro conditions. The results clearly demonstrate that Ivermectin is effective in inhibiting the development of C. irritans from the protomont to tomont stage, as well as in killing theronts – the most infectious stage in the parasite’s life cycle. At concentrations of ≥ 0.16 ppm, Ivermectin almost completely inhibited tomont development and achieved 100% theront mortality after 60 minutes of exposure, indicating a significant effect of this compound. These findings suggest the promising potential of Ivermectin as a control agent against marine fish white spot disease caused by C. irritans in marine aquaculture. However, to move towards practical application, further studies are needed to evaluate the toxicity of Ivermectin on farmed fish species, its residue persistence in fish tissues, its impact on environmental system, as well as its therapeutic efficacy when applied in pond or cage systems under farming conditions in Vietnam.
Acknowledgments
This study was funded by the collaborative research project between the ARES-CCD (Academy of Research and Higher Education, Belgium) and VNUA (Vietnam National University of Agriculture) under project number NV2025-09-O7TD.
Authors’ Contribution
Conceptualization: Anh M. Kim. Methodology: Anh M. Kim, Manh D. Vu. Formal Analysis: Anh M. Kim, Manh D. Vu, Hung M. Nguyen. Investigation: Anh M. Kim, Manh D. Vu, Hung M. Nguyen. Writing – original draft: Anh M. Kim. Writing – review & editing: Hoai D. Truong, Hung M. Nguyen, Van V. Kim. Supervision: Hoai D. Truong, Hung M. Nguyen.
Competing of Interest – COPE
The authors declare that they have no known competing financial or non-financial, professional, or personal conflicts that could have appeared to influence the work reported in this paper
Ethical Conduct Approval – IACUC
The study was approved by Vietnam National University of Agriculture under project number NV2025-09-O7TD. All applicable international, national, and institutional guidelines for the care and use of animals were followed.
Informed Consent Statement
All authors and institutions have confirmed this manuscript for publication.
Data Availability Statement
All are available upon reasonable request.