Introduction
In recent years, aquaculture has been growing strongly with a focus on sustainable development. Synbiotics, which combine probiotics and prebiotics, have attracted attention for their ability to enhance growth, immunity, and disease resistance in aquaculture species. Probiotics such as Lactobacillus and Bacillus maintain gut microbial balance, while prebiotics stimulate their activity. Supplementation of synbiotics, prebiotics, and probiotics improves nutrient absorption, feed efficiency, and pathogen resistance in fish and shrimp, offering a sustainable alternative to antibiotics.1 Despite challenges in stability and dosing, synbiotics represent a practical, eco-friendly strategy to boost aquaculture productivity and health in intensive farming systems.2 Among probiotics, Lactobacillus spp. are promising probiotics in shrimp farming due to their ability to produce antibacterial substances and stimulate immunity, helping suppress pathogenic Vibrio and enhance disease resistance.3 Oral administration is the primary delivery method, with studies showing that strains such as L. pentosus BD6 and L. plantarum 7-40 improve gut microbiota balance, growth, enzyme activity, immunity, and resistance to Vibrio infections in L. vannamei.4,5 Successful probiotic application depends on their survival in the shrimp gut, making the isolation of host-adapted Lactobacillus strains a valuable approach for Vibriosis control.
Natural prebiotics such as mannan oligosaccharides (MOS), fructo-oligosaccharides (FOS), chitosan, galacto-oligosaccharides (GOS), inulin, and various plant-derived polysaccharides have received increasing attention in aquaculture due to their ability to promote gut health, growth performance, immunity, and disease resistance.6 The supplementation of MOS in fish diets has been widely studied; for instance, dietary MOS enhanced growth performance, feed efficiency, lysozyme activity, and complement system function in Nile tilapia (Oreochromis niloticus)7 and promoted immunity in common carp (Cyprinus carpio).8 Inulin, a plant-derived fructan, has been shown to modulate gut microbiota by stimulating lactic acid bacteria, increasing short-chain fatty acid production, and improving feed utilization in carp and tilapia, thereby enhancing disease resistance under high-carbohydrate diets.9,10 Similarly, FOS are effective modulators of gut health in crustacean species. For example, in Litopenaeus vannamei, dietary inclusion of FOS significantly improved survival, phenoloxidase and lysozyme activity, and reduced Vibrio and Pseudomonas abundance in the intestine.11,12 In addition, galacto-oligosaccharides (GOS) in combination with Lactobacillus plantarum enhanced growth, digestive enzyme activities, immunity, and resistance to Vibrio alginolyticus infection in shrimp.5 More recently, chitosan and its derivatives, such as chitosan oligosaccharides (COS), have demonstrated strong immunostimulatory and antimicrobial effects, including increased hemocyte counts, antioxidant enzyme activity, and suppression of pathogenic Vibrio in shrimp intestines.13 These findings demonstrate that natural prebiotics selectively stimulate beneficial microbes while suppressing pathogens, improve nutrient absorption and immune defenses, and represent a sustainable, eco-friendly alternative to antibiotics for enhancing disease control and productivity in aquaculture.
Banana (Musa spp.) extract is recognized as a promising source of natural prebiotics due to its richness in non-digestible carbohydrates, such as resistant starch, fructooligosaccharides (FOS), and inulin-type compounds.14,15 These bioactive components resist hydrolysis by host digestive enzymes and selectively stimulate the growth and metabolic activity of beneficial gut microbiota, particularly lactic acid bacteria. In aquaculture, the inclusion of banana extract in feed has been suggested to improve nutrient absorption and digestive efficiency by creating a favorable intestinal environment, thereby enhancing immune responses and disease resistance.16–18 Furthermore, banana extract contains bioactive phytochemicals, including polyphenols and antioxidants, that may provide additional health benefits beyond its prebiotic role, contributing to improved growth performance, stress tolerance, and survival of cultured species, including Litopenaeus vannamei.19 However, few studies have investigated the effects of synbiotics on shrimp. The purpose of this study is to focus on the isolation, screening, and application of an indigenous Lactiplantibacillus plantarum strain derived from the intestine of whiteleg shrimp, combined with banana (Musa spp.) extract as a natural prebiotic. In addition, this study integrates systematic in vitro characterization (antibacterial activity and extracellular enzyme production) with in vivo evaluation of growth performance and digestive enzyme activity, thereby linking probiotic functional traits to physiological responses in shrimp. Compared with earlier banana- or L. plantarum-based synbiotic studies, including our previous work, the present study provides new evidence on the synergistic effects of a host-adapted LAB strain and banana extract, with particular emphasis on modulation of digestive enzymes and feed utilization efficiency.
Materials and Methods
Experiment 1. Isolation and evaluation of the probiotic activity of Lactobacillus species
Sampling: Healthy whiteleg shrimp, L. vannamei (7–10 g), were collected from extensive ponds in Ca Mau province. All samples were collected, placed in separate zipper bags, stored on ice, and transported to the Aqua Probiotic Lab at Can Tho University for bacterial isolation.
Isolation and identification of bacteria: Shrimp were washed and disinfected with 70% ethanol. Then, the entire intestinal tract was aseptically dissected using sterile forceps and scissors and homogenized in sterile saline solution (0.9% NaCl). The homogenates were serially diluted (10⁻¹ to 10⁻³) in saline solution, and 100 μL of each dilution was spread onto MRS agar plates supplemented with 1% CaCO₃. Plates were incubated at 37℃ under anaerobic conditions for 24–48 h.5 After incubation, distinct colonies were purified by streaking onto MRS plates. The purified strains were then cultured in MRS broth at 37°C for 24 h and preserved at –80°C a cryoprotective solution (glycerol and skim milk) for further assays.5
Antimicrobial activity: Lactobacillus isolates were tested against Vibrio parahaemolyticus using the agar well diffusion method. Cell-free supernatants (100 μL) were placed in 6 mm wells on MHA plates seeded with the pathogen (10⁷–10⁸ CFU/mL) and incubated at 30°C for 24 h. Inhibition zones were measured and classified as strong (>6 mm), medium (3–6 mm), or weak (0–3 mm).20
Extracellular enzyme assay: The cell-free supernatant of isolated bacteria was used to determine extracellular enzyme activities. Protease activity was assayed as described by Huynh et al.5 using casein as the substrate. Briefly, 100 µL of cell-free supernatant was incubated with 100 µL of 1% casein in Tris-HCl buffer (pH 7.0) at 37°C for 10 min; the reaction was stopped with 5% trichloroacetic acid, centrifuged, and enzyme activity was quantified by a modified Lowry’s method. Alpha-amylase activity was measured by the starch–iodine method.21 The assay mixture contained starch solution (0.03 g L⁻¹ in phosphate buffer, pH 7.0) and enzyme, incubated at 50°C for 30 min, then terminated with H₂SO₄ and iodine reagent, with absorbance read at 620 nm. Leu-aminopeptidase activity was determined according to Ezquerra et al.22 by incubating bacterial suspension with 1 mM p-nitroaniline in phosphate buffer (pH 7.2) at 37°C and monitoring absorbance at 410 nm for 1 h.
Identification of isolated strains by 16S rRNA gene sequencing: One potential isolate exhibiting high antibacterial and extracellular enzyme activities was identified by 16S rRNA gene sequencing (Nam Khoa Biotek Co. Ltd., Ho Chi Minh City, Vietnam).
Safety evaluation of probiotic isolate: Lactiplantibacillus plantarum LAB8.1CM was cultured, adjusted to 10⁸ CFU/mL, and sprayed onto commercial feed to prepare diets containing 10⁷–10⁹ CFU/kg. The feeds were dried (~10% moisture) and stored at 4°C until use.23
Ten healthy shrimp (~0.9 g) were stocked in 100 L tanks (10‰ salinity) with continuous aeration. Four diet treatments (basal and probiotic-supplemented at 10⁷, 10⁸, and 10⁹ CFU/kg) were tested in triplicate. Water quality was monitored twice weekly.24 Shrimp were fed 3–5% body weight twice daily for 14 days, after which the survival and LAB intestinal counts were determined.
Experiment 2. Effects of synbiotics on growth performance, digestive enzyme activity and survival of whiteleg shrimp
Experimental diets: The probiotic Lactiplantibacillus plantarum LAB8.1CM, isolated from whiteleg shrimp, was reactivated in MRS medium, freeze-dried, and mixed with skim milk protein as a carrier to obtain a concentration of 10⁸ CFU/g. The preparation was stored at 4°C until use in shrimp feed.
Banana prebiotic extract was prepared following Boonmee & Rengpipate (2015). Ripe bananas were washed, peeled, sliced, ground in hot water, and centrifuged. The supernatant was filtered, residues re-extracted, and filtrates combined before concentration by rotary evaporation. The extract was freeze-dried, ground into fine powder, and stored at −20°C until use.
A commercial shrimp diet (39-40% crude protein, 4% fat, and 5% fiber) was supplemented with L. plantarum (10⁸ CFU/g) and banana extract (BE) by spraying their saline suspensions onto pellets, followed by drying at 37°C and coating with 5% CMC to reduce leaching. The LAB concentration in feeds was verified by colony counting. The same spraying, drying, and CMC-coating procedures were applied to the control diet, using saline only. Prepared diets were stored at −20°C, with new batches produced weekly to maintain probiotic viability.
Experimental setup: A total of 100 healthy shrimp (0.69 ± 0.07 g) were acclimated for 2 weeks, then randomly distributed into fifteen 0.5 m³ tanks (five treatments, triplicates). The treatments were: (1) control diet (CtrL), (2) banana extract (0.2%, PrE), (3) L. plantarum LAB8.1CM (10⁸ CFU/kg, PrO), (4) BE 0.2% + LAB (SyN-0.2%), and (5) BE 0.4% + LAB (SyN-0.4%). Shrimp were fed experimental diets for 60 days at 3–6% body weight, divided into three meals daily. Uneaten feed was collected, dried, and used to calculate the feed conversion ratio (FCR). Tanks were siphoned every 2 days to remove waste, and water was replenished to maintain rearing conditions.
Growth parameters: At the end of the 60-day culture, the shrimp survival rate (SR) was determined. Growth performance was evaluated based on final weight (g) and calculated indices, including weight gain (WG), daily weight gain (DWG), specific growth rate (SGR), and feed conversion ratio (FCR), as described by Niu et al.25
Digestive enzyme activity: After the 60-day rearing period, five shrimp from each treatment were randomly selected and transferred to glass tanks with water at the same salinity as the rearing conditions. Shrimp were starved for 24 h prior to sampling. The midgut and hepatopancreas were then dissected, and each sample was placed in a sterile Eppendorf tube containing 700 µL of phosphate-buffered saline (PBS). Samples were homogenized and centrifuged at 3000 rpm for 10 min at 4 °C, and the resulting supernatant was used to assess digestive enzyme activity. Alpha-amylase, protease, and leu-aminopeptidase were analyzed as described above. β-galactosidase activity was determined as described by Vinderola and Reinheimer26 with minor modifications. Briefly, 100 µL of the sample was mixed with 900 µL of PBS and 200 µL of o-nitrophenyl-β-D-galactopyranoside (ONPG, 4 mg/mL). The reaction mixtures were incubated in a water bath at 37 °C for 15 min, after which 0.5 mL of 1 M Na₂CO₃ was added to stop the reaction. Optical densities were measured at 420 nm and 560 nm, and enzyme activity was expressed in units of U/mg protein. Total protein concentration was determined by the Bradford method using bovine serum albumin as the standard.27 Each treatment was analyzed using five shrimp.
Statistical analysis
Data are expressed as mean ± standard error (SE). Statistical analyses were performed using IBM SPSS v22.0. One-way ANOVA was applied to assess differences among treatment means, and Tukey’s post hoc test was used to identify significant differences at a 95% confidence level (p<0.05).
Results
Experiment 1. Isolation and evaluation of the probiotic activity of Lactobacillus species
Isolation and identification of bacteria: A total of 249 strains producing clear zones on MRS agar supplemented with CaCO₃ were isolated. After purification, colonies were predominantly white to yellowish, round (2–3 mm in diameter), smooth, and entire. Microscopic characterization showed that 15 of these strains were Gram-positive, with 60% being rod-shaped and 40% spherical. All were catalase- and oxidase-negative, non-motile, and non-spore-forming (Table 1).
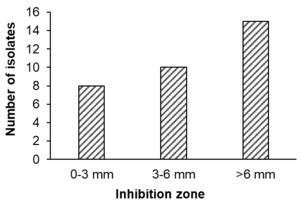
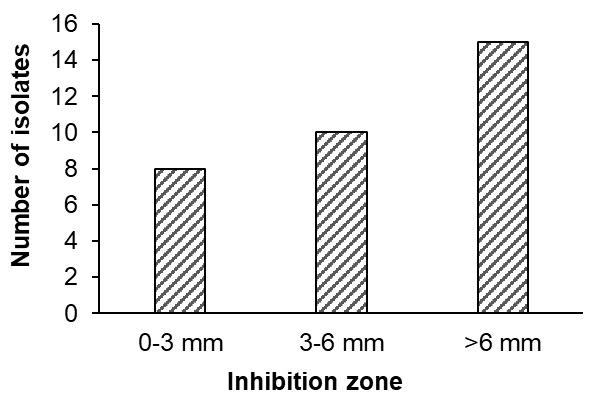
Antibacterial activity: A total of 33 out of 249 isolates demonstrated inhibitory activity against Vibrio parahaemolyticus, with inhibition zone diameters ranging from 2 to 12 mm. Based on the criteria proposed by Pan et al.,20 8 isolates (24.2%) exhibited weak inhibition, 10 isolates (30.3%) showed moderate inhibition, and 15 isolates (45.5%) were classified as strong inhibitors (Figure 1).
Production of extracellular enzymes: The strain LAB8.1CM had the highest protease activity and significantly compared the others (p < 0.05). Similarly, strains LAB1.2CM and LAB8.1CM showed the highest Leu-aminopeptidase activity and were significantly higher than the others (p < 0.05). However, these two strains displayed the lowest activity, being significantly different compared to LAB1.2ST and LAB6.1ST (Table 2).
Identification of isolated strains by 16S rRNA gene sequencing: the strains LAB8.1CM and LAB1.2CM exhibited 99.9% similarity with Lactiplantibacillus plantarum (formerly known as Lactobacillus plantarum).
Safety evaluation of probiotic isolates: After 14 days of the trial, no significant differences in water quality parameters were observed among the experimental diets (p > 0.05) (Table 3). LAB counts in shrimp fed probiotics were significantly higher (p < 0.05) than those in the basal diet (Table 4). The levels increased with probiotic dosage, with the highest values observed at 10⁸ and 10⁹ CFU/kg, in which no significant difference was found between these treatments (p > 0.05). No significant difference in survival rate was observed among all treatments (p > 0.05).
Experiment 2. Effects of synbiotic on growth performance, digestive enzyme activity and survival of whiteleg shrimp
Water quality parameters: The measured water quality parameters are presented in Table 4. No significant differences (p > 0.05) were observed among the experimental and control groups for temperature, pH, salinity, dissolved oxygen (DO), total ammonia nitrogen (TAN), nitrite (N-NO₂⁻), and phosphate (P-PO₄³⁻).
Growth performance and survival: Dietary supplementation with 0.2% synbiotic significantly improved (p < 0.05) growth performance of whiteleg shrimp compared with the control (Table 5). Final weight, WG, DWG, and SGR were significantly higher in the SyN-0.2% group than in the control, while no significant differences (p > 0.05) were found among the experimental diets (PrE, PrO, SyN-0.2%, SyN-0.4%). Survival did not differ among treatments (p > 0.05). Feed conversion ratio (FCR) was significantly reduced (p < 0.05) in synbiotic-supplemented groups, with the greatest improvement in SyN-0.2%.
Digestive enzyme activity: Dietary probiotics, prebiotics, and synbiotics enhanced digestive enzyme activities in shrimp (Table 6). Synbiotic diets significantly increased (p < 0.05) intestinal α-amylase and β-galactosidase activities, as well as protease and leucine aminopeptidase activities in both the hepatopancreas and intestine, compared with the control. However, no significant effects (p > 0.05) were observed in shrimp fed PrE and PrO diets.
Discussion
The application of probiotics, prebiotics, and synbiotics in aquaculture has recently emerged as a sustainable alternative to antibiotics, contributing to enhanced animal health and productivity. Probiotics, indigenous bacterial strains isolated from the host organism itself, are particularly valued for their greater effectiveness compared to those derived from other sources.28 Therefore, the development of native probiotics is essential for advancing sustainable aquaculture practices. In the present study, 33 out of 249 isolates showed antibacterial activity, with inhibition zones above 2 mm. Among them, the highest antimicrobial activity against V. parahaemolyticus was observed with LAB1.2ST, LAB6.1ST, LAB1.2CM, and LAB8.1CM isolates, whereas the remaining isolates exhibited the lowest antimicrobial activity (p < 0.05). A study by Dung et al.29 reported that eight LAB strains exhibited resistance against pathogenic Vibrio parahaemolyticus, the causative agent of early mortality syndrome (EMS) in cultured shrimp, with inhibition zones ranging from 2.6 to 4.17 mm. Similarly, Le et al.30 found that 5 of 16 LAB strains isolated from white shrimp intestines inhibited Vibrio pathogens, including V. parahaemolyticus, with inhibition zones ranging from 5 to 10 mm. In addition, the ability to secrete extracellular enzymes is a critical criterion for selecting beneficial bacteria, as it supports nutrient utilization and host health in aquaculture. The highest protease and leu-aminopeptidase activity was observed in LAB1.2CM and LAB8.1CM isolates, while the lowest activity was in LAB1.2ST and LAB6.1ST isolates, respectively. These enzymatic activities are important for enhancing nutrient utilization and probiotic efficacy (e.g., through enhancing protein and amino acid digestibility, stimulating the expression of host peptidases, and modulating the proteolytic activity of gut microbes).31,32 Based on the findings from antimicrobial activity and enzyme activity, the isolates LAB8.1CM and LAB1.2CM were selected for molecular identification, showing high antibacterial activity and extracellular enzymes. Moreover, the LAB8.1CM strain was shown to be safe and suitable for use as a probiotic in shrimp.
Several studies have demonstrated the effectiveness of synbiotics containing natural extracts and probiotics (Lactobacillus spp., Bacillus spp.) through the diet in promoting growth performance in aquatic species.33,34 In this study, banana was used as a prebiotic due to its high nutritional content (carbohydrates, minerals, and vitamins), easy availability, and low market price in Vietnam.35 Santo et al.36 reported that banana could promote the growth of L. acidophilus L10. Shrimp fed the SyN-0.2% diet exhibited the highest WG, SGR, DWG and best FCR, suggesting the synergistic effect of combined L. plantarum LAB8.1CM and banana extract. Similarly, the dietary inclusion of synbiotics from L. plantarum CMT1 plus noni fruit extract significantly improved the growth performance and FCR of the whiteleg shrimp.37 In addition, Boonmee and Rengpipat38 reported that the combined addition of B. subtilis S1 and ripe banana extract to diets resulted in significantly greater growth performance in L. vannamei than the individual additions. The growth enhancement observed with synbiotics in this study is evident, and a synergistic mechanism of action between probiotics and prebiotics has been proposed. Prebiotics, such as banana extract, can selectively stimulate the growth and activity of probiotics, while probiotics, such as L. plantarum LAB8.1CM, produce exogenous enzymes (e.g., proteases, amylases, and aminopeptidase) that enhance the breakdown of complex macromolecules into absorbable nutrients. This improved digestive enzyme activity has been linked to better feed utilization and growth performance in aquatic animals, including shrimp.39,40 Indeed, the administration of 0.2% synbiotics increased digestive enzyme activities of whiteleg shrimp, including protease, leu-aminopeptidase, and β-galactosidase, compared to the control diet. This is consistent with the findings of Pardede et al.,41 who demonstrated that the use of synbiotics in the shrimp diet could enhance digestive enzyme activity. Moreover, the fermentation of the prebiotic components in the banana extract (e.g., carbohydrate, vitamin, mineral) by probiotic, L. plantarum LAB8.1CM, not only stimulated the proliferation of beneficial bacteria but also suppressed the growth of potentially pathogenic microorganisms in the intestine, thereby contributing to promoting growth performance.42 In the synergistic effect of SYN, both the probiotic dose and the dose/type of prebiotics can significantly influence shrimp growth and health. Indeed, our observations on improving growth performance did not relate to increasing the dose of banana extract.
In conclusion, the present study demonstrated that the isolated L. plantarum LAB8.1CM from shrimp and the banana extract functioned effectively as a probiotic and prebiotic, respectively. Subsequently, a synbiotic formulation was developed using the selected combination of L. plantarum LAB8.1CM at 108 CFU/kg and 0.2% banana extract. This synbiotic formulation exhibited beneficial effects on the growth performance and digestive enzyme of whiteleg shrimp, which served as the in vivo model organism. These results suggest that the combination of L. plantarum LAB8.1CM and banana extract represents a promising synbiotic candidate for aquaculture species. Further research is necessary to evaluate the effectiveness of the synbiotic on the disease resistance and immune response of whiteleg shrimp, thereby providing essential information for its application in aquaculture farms.
Acknowledgments
This study was sponsored by the Vietnamese Ministry of Education and Training for scientific research project (the project code B2024-TCT-03).
Authors’ Contribution
Conceptualization: Giang T. Huynh (Equal). Methodology: Giang T. Huynh (Equal), Ha T.K. Nguyen (Equal), Huong T.T. Do (Equal), Tu T.C. Phan (Equal). Formal Analysis: Giang T. Huynh (Equal), Hai H. Vu (Equal). Writing – original draft: Giang T. Huynh (Lead). Writing – review & editing: Giang T. Huynh (Equal), Tu T.C. Phan (Equal). Funding acquisition: Giang T. Huynh (Lead).
Ethical Conduct Approval – IACUC
This experiment was carried out in accordance with national guidelines on the protection of animals and experimental animal welfare in Vietnam (Law on Animal Health, 2015, Vietnam National Assembly, No. 79/2015/QH13).
Competing of Interest – COPE
No competing interests were disclosed.
Informed Consent Statement
All authors and institutions have confirmed this manuscript for publication.
Data Availability Statement
All are available upon reasonable request.