1. Introduction
The accelerated loss of biodiversity represents one of the most formidable challenges humanities currently faces. Factors such as biological characteristics, environmental pollution, and other human activities are associated with increased extinction risk for many species, with some pushed to the brink of extinction.1 However, the risk of extinction is not uniformly distributed across species.2 This phenomenon, known as ‘non-random extinction’ or extinction risk selectivity, is observed not only in extant taxa but also in paleobiological patterns.3 Selective extinction of specific species accelerates biodiversity loss, potentially eradicating entire branches of the evolutionary tree along with their unique evolutionary histories.4,5
Schizothoracinae, a subfamily of Cyprinidae, is endemic to the rivers and lakes of the Tibetan Plateau and faces a severe conservation crisis, with over half of its species threatened.6 Their evolutionary history is deeply intertwined with the plateau’s dynamic environment.6–9 As the only cyprinid group adapted to this extreme setting, they have evolved specialized traits, including a strong reliance on rapids and high-gradient river flows (rheophily).10 This very adaptation, however, underlies their increased risk of extinction. Rheophilic species like these are intrinsically vulnerable because their suitable habitats—rapids—are naturally fragmented, isolating populations into small, distinct patches, which leads to significant genetic structuring and a high susceptibility to demographic and environmental stochasticity, including bottleneck effects.11 Consequently, their heightened association with environmental conditions, coupled with their fragmented distribution, makes them ideal model organisms for studying species-environment correlations and critically important for maintaining the ecological integrity and biodiversity of the Tibetan Plateau’s aquatic ecosystems.6 China, a nation rich in biodiversity, has its freshwater ecosystems particularly under scrutiny. The “China Red List of Biodiversity,” co-compiled by the Ministry of Ecology and Environment and the Chinese Academy of Sciences, documents 1,591 species of freshwater fish (excluding alien species) in China, which are distributed across 18 orders, 51 families, and 328 genera.12 Yet, influenced by inherent biological characteristics of freshwater fishes and human activities, these species face escalating extinction risks.7
Previous studies have revealed the ecological functions of freshwater fishes. Investigations have ranged from the biodiversity patterns of the Yangtze River basin13 to the calculation of extinction risk indices for endemic species in the upper Yangtze,14 and studies on the relationship between species richness, endemism, and threatened species in Chinese freshwater fishes.15,16 The assessment of the threatened status and causes affecting inland fish, as outlined in the Red List,17 the analysis of fish resources in the Yangtze River basin alongside conservation strategies for various sections,18 and the systematic examination of the relationship between species richness, endemic species, genera, and ecological factors in six plateau lakes in central Yunnan have all significantly advanced our understanding of these issues.16
However, in evaluating the threatened status of freshwater fish species within China, these studies have not sufficiently accounted for the implications of the Phylogenetic Isolation Index (PII). PII is a crucial metric that quantifies the phylogenetic distinctiveness of a species, reflecting its evolutionary uniqueness and the extent of its isolation from their closest relatives.19,20 Species with high PII values represent a unique genetic and evolutionary history and are therefore prioritized for conservation to prevent significant biodiversity loss.19 Additionally, while Cao et al.17 documented threatened species in major families, they did not quantify phylogenetic uniqueness or test the statistical significance of extinction risk bias. Our study fills this gap by quantifying the statistical significance of this bias and identifying families with anomalously threatening proportions that were not statistically validated in prior analyses.
Previous research has quantitatively analyzed the relationship between biological characteristics and extinction risk through integrated ecological and conservation approaches, providing key scientific bases for understanding the mechanisms of species extinction and formulating scientifically sound conservation measures.21 Despite significant progress in this field, the correlation between extinction risk and biological traits may vary for specific species and taxonomic groups. For example, endangered birds threatened by invasive species typically have longer metabolic cycles and larger sizes, while those facing habitat loss have specialized habitats and smaller sizes 22. Previous trait–extinction risk analyses (e.g., Xing et al.15) have focused on lowland freshwater fishes, but the drivers of risk for plateau-adapted groups, such as Schizothoracinae, remain unclear. Additionally, PII and HFI have not been integrated into a unified assessment framework for Chinese freshwater fishes.
In view of this, our research provides an extensive analysis of the conservation imperatives for Chinese freshwater fishes by examining their PII and evaluating their vulnerability to threats. We focus on using the phylogenetic tree of Chinese freshwater fishes to explore the PII of these species, and we analyze whether extinction risks are randomly distributed among them. Additionally, the study methodically quantifies the biological characteristics of the Schizothoracinae subfamily, aiming to explain potential connections between these traits and their risk of extinction. Building upon this foundation, our investigation extends to incorporate the Human Footprint Index (HFI) 23 in conjunction with the conservation status classification of the Schizothoracinae subfamily. This integrative approach enables a more comprehensive understanding of the interplay between human activities and the conservation status of these species.
Consequently, our study provides quantitative support for future conservation strategies and management decisions. This study represents the first comprehensive integration of the phylogenetic isolation index (PII), extinction risk non-randomness, and human disturbance to assess conservation priorities for Chinese freshwater fish.
2. Materials and Methods
2.1. Data Sources
This study is based on China’s Red List of Biodiversity,12 which provides a comprehensive list of freshwater fish species in China, along with their assessment levels, compiled from a combination of specimens, literature, and expert information. While the IUCN Red List of Threatened Species24 is a widely recognized database, the Chinese Red List focuses on protected fish in China, providing more local and relevant insights into species conservation in the region. This study also synthesizes information from various publications, reports, and multiple databases (e.g., FishBase,25 Taiwan Fish Database,26 which are instrumental in exploring the biological characteristics of the Schizothoracinae subfamily and understanding the relationship between their habitat features and extinction risk.
2.1.1. Phylogeny of freshwater fishes from China
In this study, we constructed a phylogenetic tree that encompasses all 1,591 species of freshwater fishes from “China’s Red List of Biodiversity”. This was achieved by utilizing the R package “FishPhyloMaker”,27 which facilitated the assembly of the phylogenetic structure based on comprehensive taxonomic data.
2.1.2. Compilation of Freshwater Fish Species List and Classification of Conservation Status
The list assesses the extinction risk of Chinese freshwater fishes according to the categories and criteria of the IUCN Red List. In the IUCN Red List, species classified as Critically Endangered (CR), Endangered (EN), and Vulnerable (VU) are collectively referred to as threatened species. Of the 1591 assessed species, 358 are categorized as threatened, while 613 are considered non-threatened. For subsequent quantitative analysis, this study categorizes freshwater fish species by conservation status as follows: Least Concern (LC) = 0, Near Threatened (NT) = 1, Vulnerable (VU) = 2, Endangered (EN) = 3, Critically Endangered (CR) = 4, Extinct (EX) or Regionally Extinct (RE) = 5.
2.1.3. Collection of Information on Schizothoracinae subfamily Fish
There are 12 genera and 125 species in the Schizothoracinae subfamily.6We collected various information about Schizothoracinae subfamily,6,17 including maximum body length, dispersal distance, sexual maturity age, egg diameter, initial fry length, spawning quantity, breeding water temperature, altitude, and geographical coordinates (latitude and longitude).27 Additionally, we quantified several key biological parameters,15,17 such as river flow velocity (slow = 1, moderate = 2, fast = 3), climate zone (temperate = 1, subtropical = 2, tropical = 3), diet (omnivorous = 1, carnivorous = 2, herbivorous = 3), spawning season (spring/summer = 1, autumn/winter = 2, summer/autumn = 3), egg color (light grey = 1, light yellow = 2, yellow = 3, golden yellow = 4, orange-red = 5), and habitat type (upper/middle layer = 1, bottom layer = 2, lower/middle layer = 3).
In analyzing the correlation between changes in the Human Footprint Index (HFI) on the Tibetan Plateau and the conservation status of the Schizothoracinae subfamily, we primarily utilized the Tibetan Plateau Human Footprint dataset from 1990 to 2017.28 This dataset incorporates spatial data that represents various human activities, including population density, land use, grazing density, night lights, railways, and roads.
2.2. Data Analysis
2.2.1. PII of Chinese freshwater fishes
In this study, phylogenetic tree analysis and isolation quantification were conducted in a Python environment. By traversing each leaf node of the phylogenetic tree, which represents different species, we utilized the distance method to calculate the distance of each species from the root of the tree. This distance was used as a metric to measure the PII of the species.20 To identify species with unique phylogenetic characteristics, we sorted the obtained data in descending order of their isolation degree.
2.2.2. Threatened Status of Chinese freshwater fishes
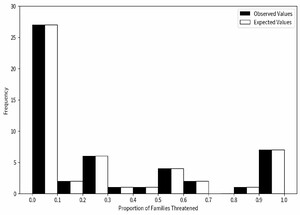
In this study, we found that 358 of the 1591 species of freshwater fishes in China are classified by IUCN as threatened. To conduct an in-depth analysis, we randomly selected 358 species from the total and recorded their taxonomic families. This random sampling method provided us with an approximate inferential means based on the sample. We performed 10,000 repeat simulations using the Python programming language to calculate the proportion of threatened species in each family and plotted a frequency histogram. This histogram was divided into ten equal intervals, each with a width of 0.1. Since the frequencies in the 0.1-0.5 and 0.5-0.9 intervals were extremely small or zero, we combined these intervals into one category. This frequency histogram shows the predicted distribution of extinction risk. If the extinction risk is randomly distributed among different families, there should be no significant difference between observed and expected values. To test this, we further employed the chi-square test (χ2) to evaluate the results, comparing the calculated chi-square value (χ2) with the degrees of freedom (df) and significance level. If the chi-square value is lower than the critical value, we reject the null hypothesis, indicating a significant difference between the observed and expected values. Conversely, if the chi-square value is higher than the critical value, we reject the null hypothesis, suggesting a significant difference between observed and expected values, thus not supporting the hypothesis of random distribution of extinction risk among families.
Additionally, we used the binomial distribution to determine which families contained anomalously high or low numbers of threatened species.29 In each family, we assumed that the status of species being threatened was random, i.e., in a family containing N species, each species had the same probability of being threatened. We calculated the probability of observing the actual number of threatened species in each family under this assumption. If the probability was less than 0.05, we considered the number of threatened species in that family to be significantly different from the expected number under the assumption of randomness.
2.2.3. Extinction Risk of Schizothoracinae Subfamily
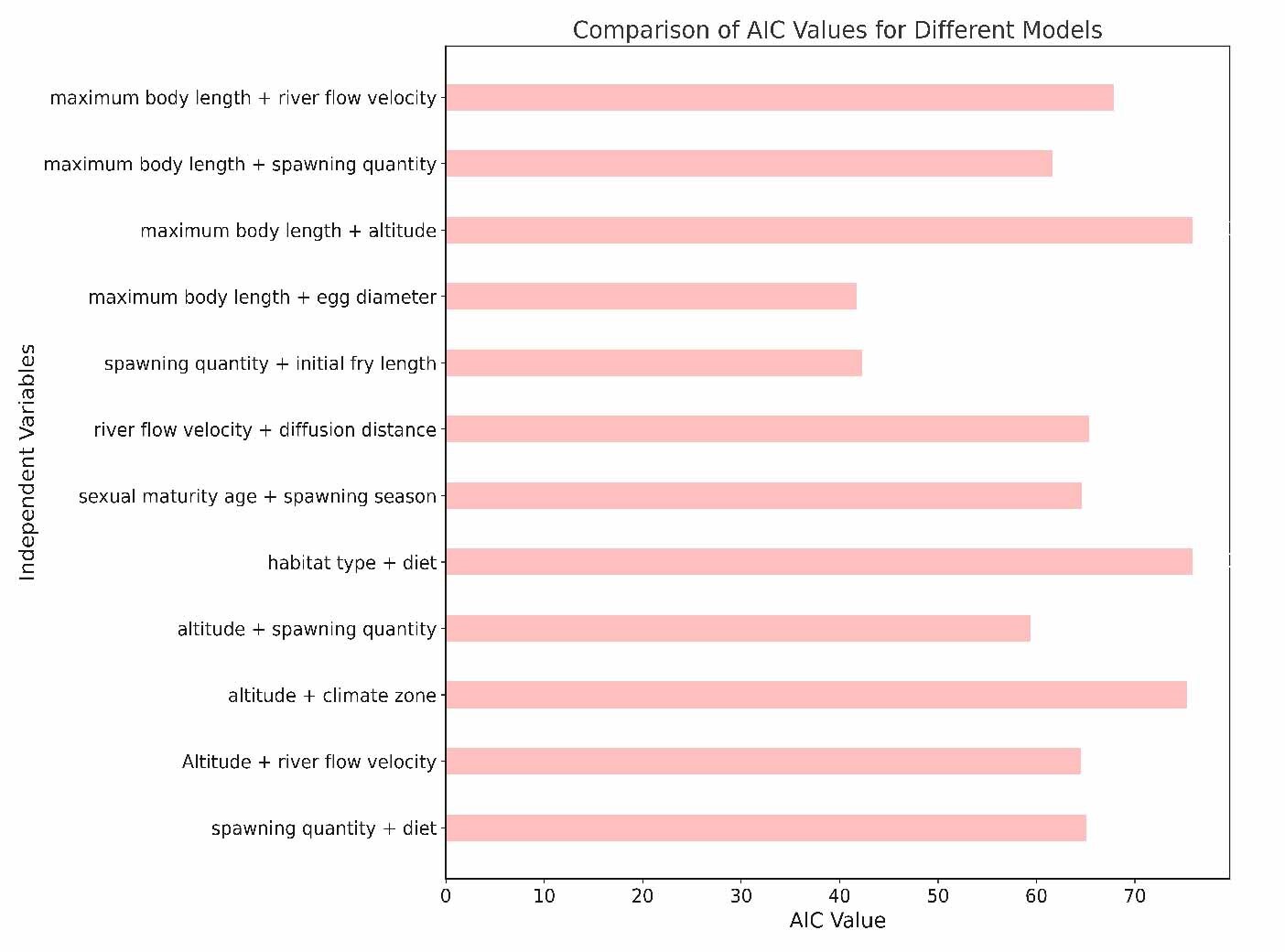
This study aims to investigate the relationship between species characteristics and the extinction risk of Schizothoracinae subfamily fish. We employed a Generalized Linear Model (GLM) for analysis. First, we categorized the extinction risk into a binary classification: species classified as Critically Endangered, Endangered, or Vulnerable were grouped as threatened species (1), while those categorized as Near Threatened or Least Concern were considered non-threatened species (0). In the GLM analysis, extinction risk was treated as the dependent variable, with other biological characteristics serving as independent variables.
Then, we utilized the binomial distribution and the logit link function for data processing and modeled all possible combinations of characteristics and their interactions. By examining the Model Summary, we obtained crucial statistical information, including coefficient estimates, standard errors, z-values, and p-values. Finally, we used the Akaike Information Criterion (AIC) as the basis for model selection. Additionally, we calculated the AIC value to assess the predictive performance of the model.29 In this study, we initially considered 62 species of the Schizothoracinae subfamily. However, not all species had complete data for the variables of interest (e.g., body length, dispersal distance, spawning quantity, dietary habits). Therefore, we adopted a complete-case analysis approach, excluding species for which essential data were lacking for each specific model. This led to variations in the number of species included across different models. We acknowledge that this approach might have implications for the sample size and could potentially introduce bias into our findings. Hence, the interpretation of our results was made with careful consideration of these limitations, and we assessed the robustness of our conclusions by examining the consistency of results across different models.
2.2.4. Relationship between HFI and Species Conservation Status
In this study, we focused on analyzing the impact of human activities in the Tibetan Plateau region on the Schizothoracinae subfamily. We assessed the trend of human activity intensity in this area by calculating changes in the HFI from 1990 to 2017. After determining the average HFI for each conservation status category, we conducted a one-way Analysis of Variance (ANOVA), with conservation status categories as the factor and HFI values as the response variable. This approach allowed us to determine whether changes in the HFI for species with different conservation statuses in the Tibetan Plateau exceeded the bounds of random variation.
3. Results
3.1. PII Analysis
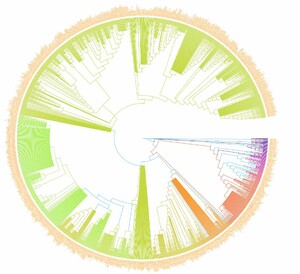
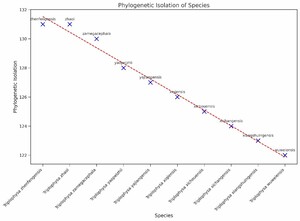
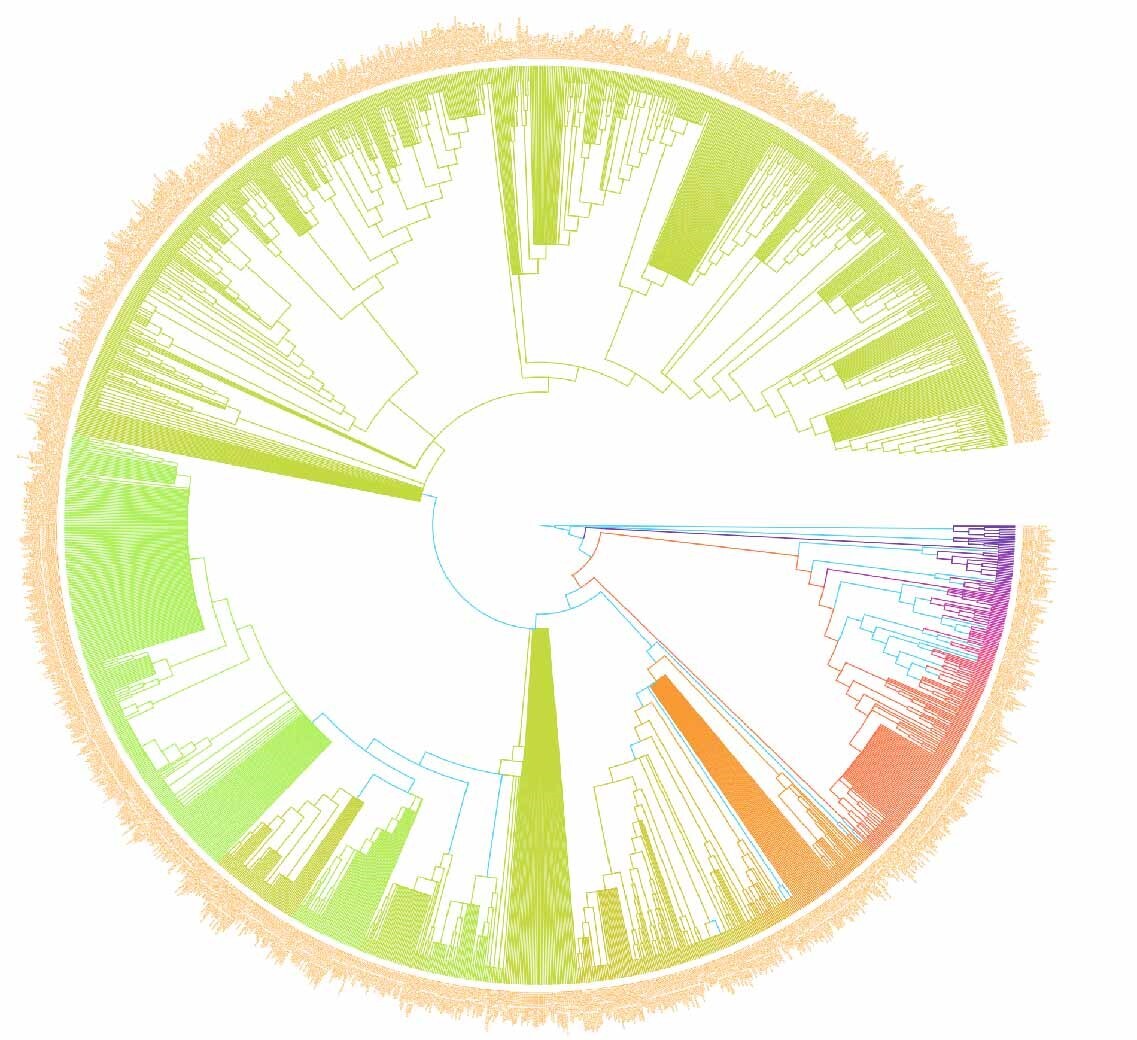
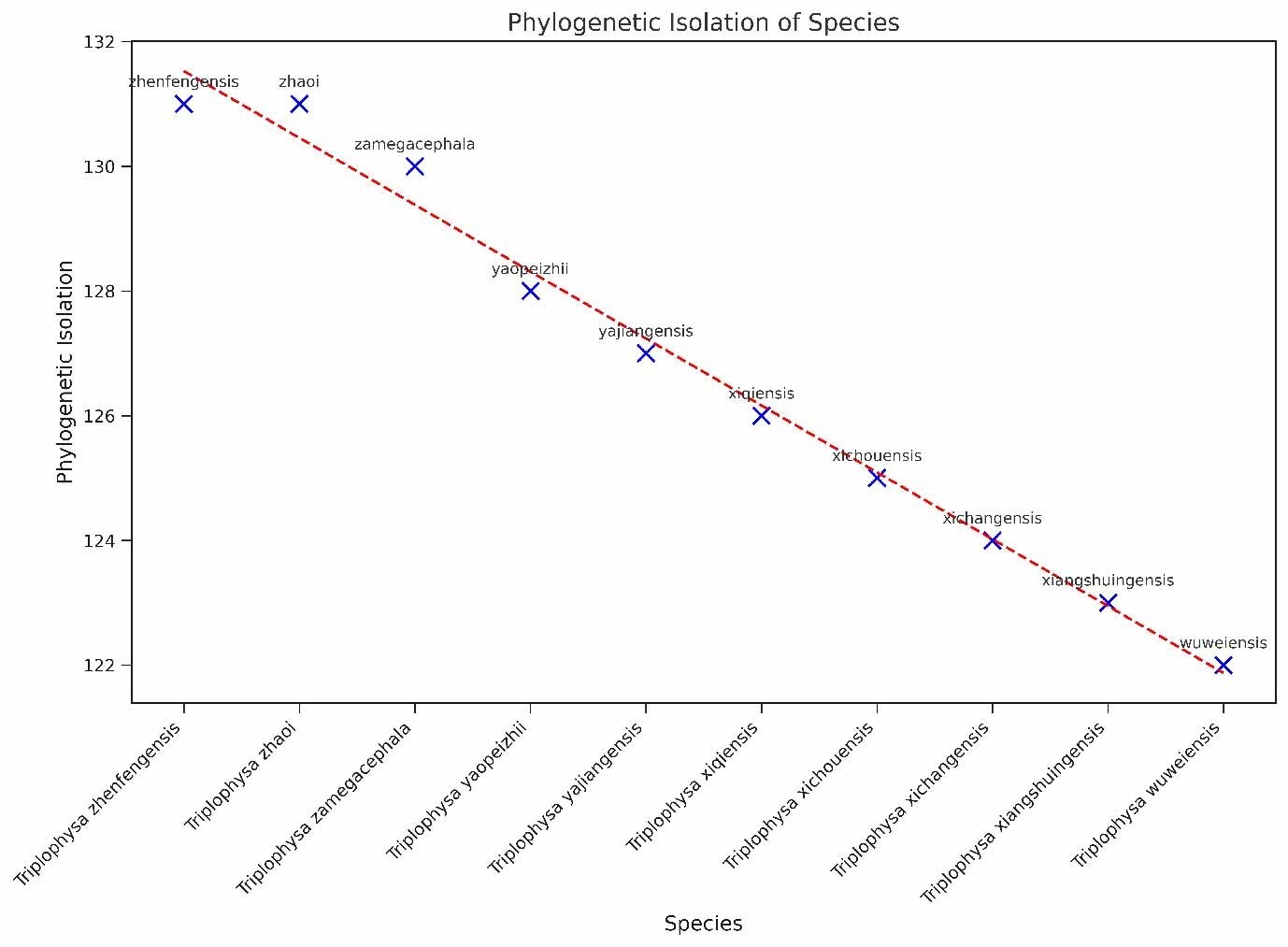
In this study, we investigated the PII of Chinese freshwater fishes by analyzing the phylogenetic tree (Figure 1). This research encompassed 1591 species of Chinese freshwater fishes, with a focus on those exhibiting significant isolation on the phylogenetic tree. The results revealed that the top ten species, i.e., Triplophysa zhenfengensis, T. zhaoi, T. zamegacephala, T. yaopeizhii, T. yajiangensis, T. xiqiensis, T. xichouensis, T. xichangensis, T. xiangshuingensis, T. wuweiensis– with the most pronounced PII–all belong to the Nemacheilidae family (Figure 2). These high-PII species in Nemacheilidae have not been prioritized in previous conservation lists (e.g., Cao et al.17), but they represent irreplaceable evolutionary lineages, emphasizing the need to incorporate phylogenetic uniqueness into conservation planning. This finding highlights the extraordinary phylogenetic uniqueness of these species within the Nemacheilidae family. The marked PII of these species may indicate that they carry unique evolutionary historical information. Given this, these species should be considered a priority in biodiversity conservation efforts. Their potential loss implies not just a reduction in species numbers but may also lead to the irreversible loss of specific evolutionary histories.
Therefore, from the perspective of biological conservation and biodiversity maintenance, special attention should be paid to these species with high PII.
3.2. Threatened Status in Freshwater Fishes
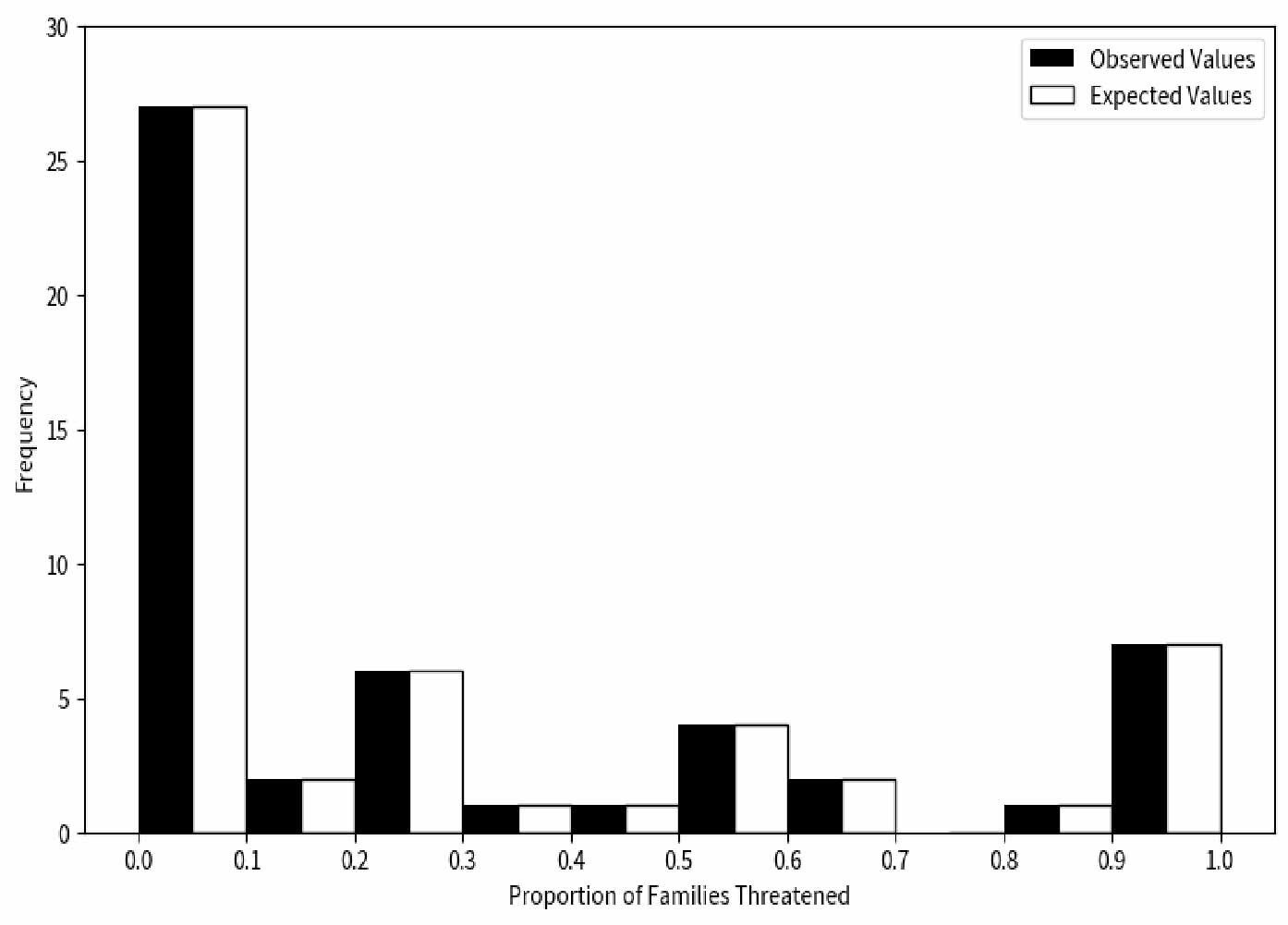
In this study, we observed the proportion of threatened species across different families through 10,000 repeated simulations. Our null hypothesis posited that there is no significant difference between the proportion of threatened species in each family and the expected values (Figure 3). Unlike descriptive reports of Cyprinidae’s high endangerment,17 our chi-square test (χ² = 10.033, df = 4, p<0.05) confirms that extinction risk is non-randomly distributed across families, with 7 families (e.g., Acipenseridae) showing statistically significant overrepresentation of threatened species (binomial test, p<1.72e-04). These findings provided sufficient grounds to reject the null hypothesis, leading to the conclusion that the extinction risk faced by species is not randomly distributed across different families but exhibits certain biases. This suggests that species in certain families may be more susceptible to extinction risk than those in other families.
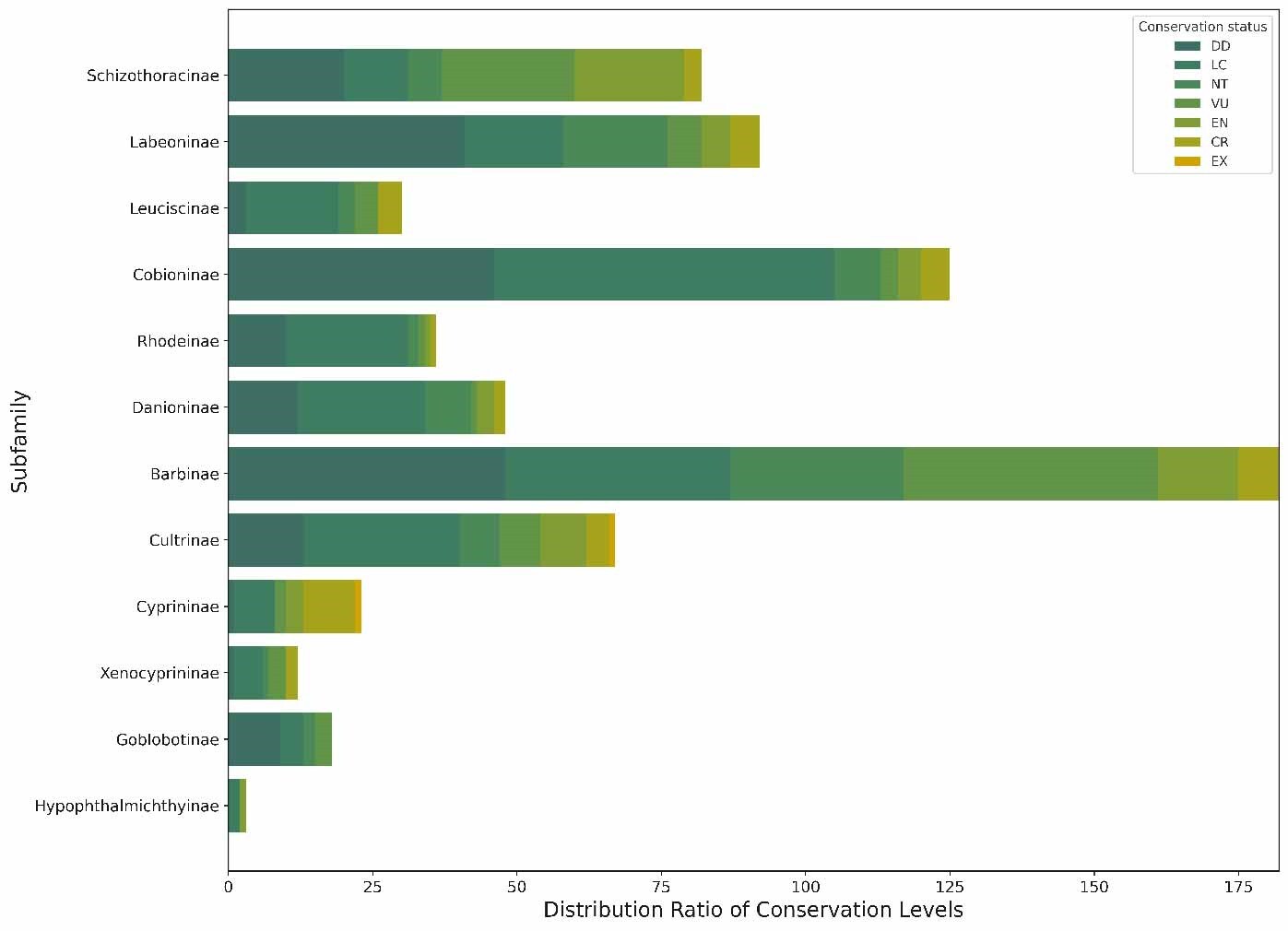
To test whether extinction risk is randomly distributed across families, we conducted a chi-square test (χ²=10.033, df=4, p<0.05), which rejected the null hypothesis—indicating that the proportion of threatened species is non-randomly distributed among families (Figure 4). Among all families, Cyprinidae had the highest number of threatened species (199 species, 55.59% of all threatened fishes), while 7 small families (e.g., Acipenseridae, Polyodontidae) had a 100% threat proportion (despite small sample sizes of 1-3 species). Furthermore, we calculated the threatened proportion and binomial probability for each family (Table 1) and conducted independent tests for each. After adjusting the critical values using the Dunn-Sidak method,30 we found that the critical values at 5% and 1% significance levels were p< 1.72e-04 and p < 3.36e-05, respectively. A p-value smaller than these thresholds indicates statistical significance. No family showed a statistically significant excess of threatened species (overrepresentation). Notably, families with small sample sizes (n ≤ 5 species) had low statistical power, so their 100% threat proportion should be interpreted as a descriptive pattern rather than statistically validated overrepresentation. These results collectively confirm non-random extinction risk distribution, driven by both underrepresentation in some families and extreme proportional threat in small families, rather than uniform randomness across all taxa.
3.3. Association between Ecological and Biological Characteristics and Extinction Risk of Schizothoracinae Subfamily
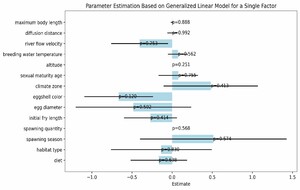
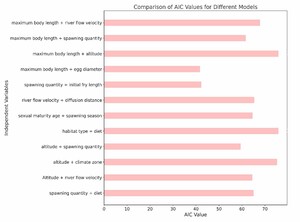
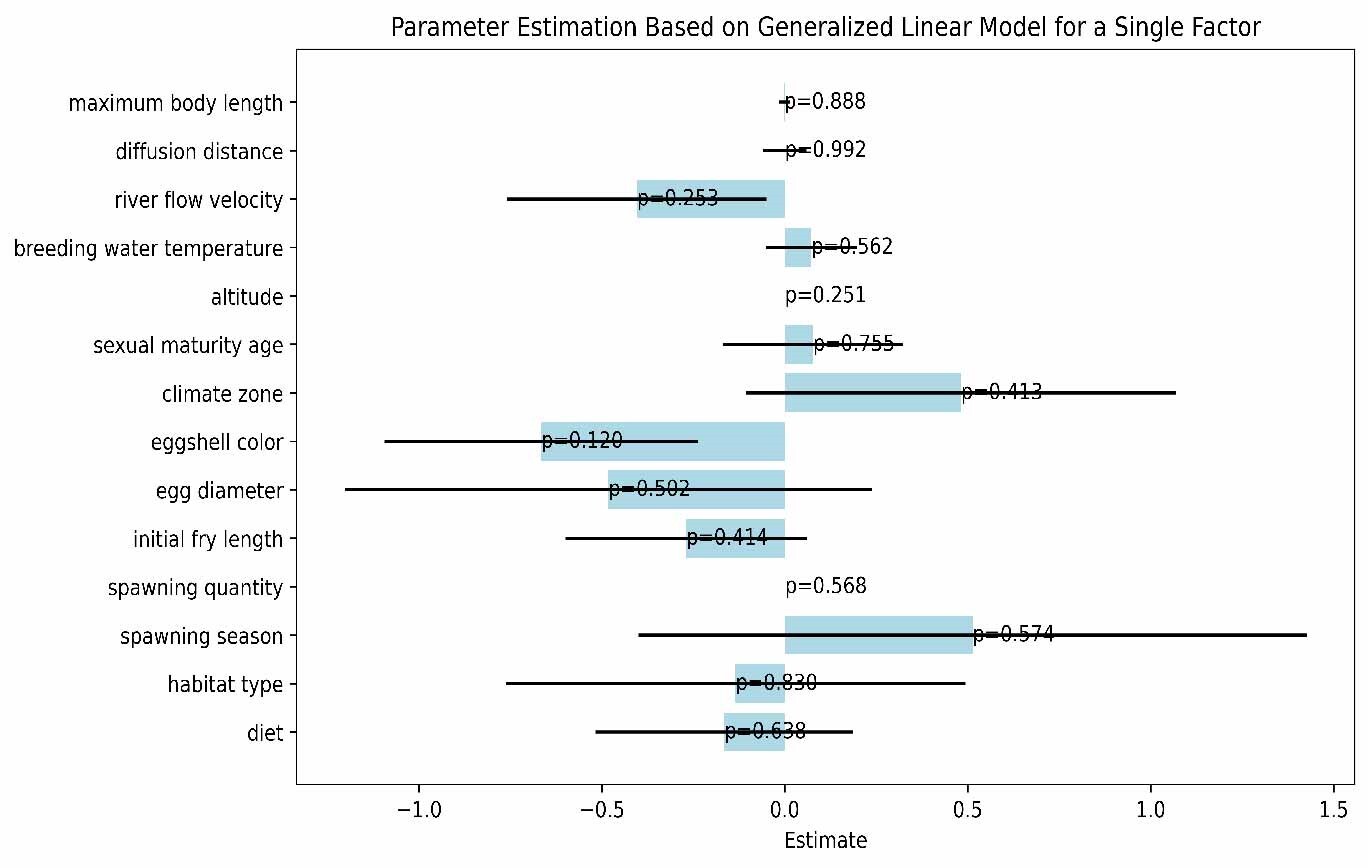
In this study, we explored the relationship between various ecological and biological characteristics of the Schizothoracinae subfamily fish and their risk of extinction. Notably, our analysis revealed no statistically significant correlations between biological traits, such as maximum body length, egg diameter, and age at sexual maturity, and the extinction risk of these freshwater fishes. Similarly, habitat characteristics like breeding water temperature and habitat altitude also appeared to be uncorrelated with extinction risk, as depicted in Figures 5 and 6. While these findings are unexpected, they provide valuable insights into biodiversity research, suggesting that the determinants of species extinction risk may be more complex and variable than previously thought. It seems that a complex interplay of various biological and environmental factors could be at play, indicating that species extinction risk is a multifaceted issue.
However, it is crucial to acknowledge the limitations of our study. The absence of significant findings could be attributed to various factors, including potential limitations in our dataset, such as sample size, or unaccounted variables that might influence extinction risk. Additionally, the methods employed may not effectively capture the interactions between different factors.
3.4. Association Between HFI and Conservation Status Categories
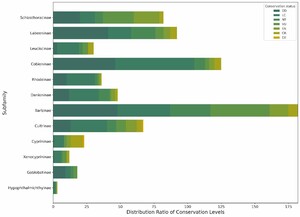
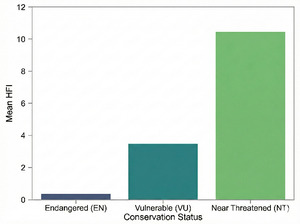
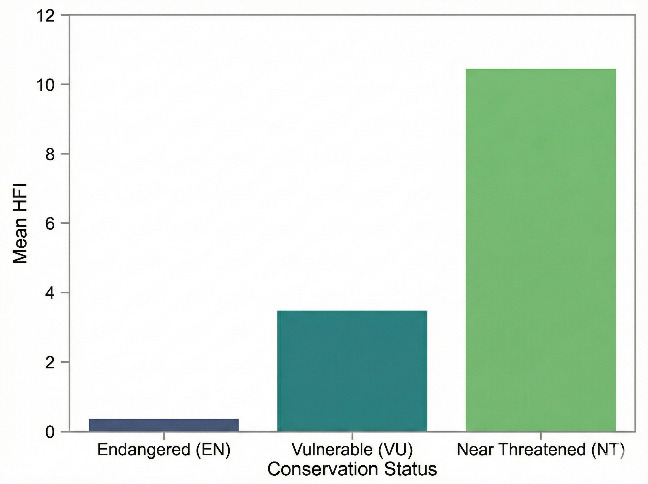
We quantitatively assessed the potential impact of human activities on the conservation status of different species within the Schizothoracinae subfamily in the Tibetan Plateau region using the HFI. Our calculations revealed that species in the Endangered (EN) category had the lowest HFI mean (µ = 0.342542), while Near Threatened (NT) species exhibited a significantly higher mean HFI (µ = 10.3667304). Vulnerable (VU) species had an intermediate mean HFI (µ = 3.464580) (Figure 7). An ANOVA test yielded an F-statistic of 9.4849 and a p-value of 0.01987, indicating that NT species are significantly associated with areas of higher human activity intensity, whereas endangered (EN) species are predominantly found in areas with lower human disturbance. These patterns suggest differences in the degree of co-occurrence between species of different conservation levels and human activities, which may reflect variations in their ecological niches rather than direct tolerance or sensitivity to disturbance. In contrast, near threatened species may exhibit a higher degree of co-occurrence with human-modified environments, which could be attributed to overlapping ecological niches (e.g., broader habitat requirements) rather than evidence of adaptive responses to disturbance (for which temporal or experimental data are lacking). Furthermore, this finding suggests that in formulating targeted conservation measures, it is essential to consider the varying responses of species to human activities. To more effectively protect these valuable aspects of biodiversity, further research into the ecological needs of these species and their adaptability to environmental changes is crucial.
4. Discussion
4.1. Threatened Status of Freshwater Fishes in China
This study investigated the threatened status of Chinese freshwater fishes, revealing a non-random distribution of extinction risk across different fish families, which supports the hypothesis of non-random distribution of extinction risk.30
Our comparison of simulated and observed data indicates a general alignment of threatened species numbers across families with the simulated predictions, suggesting that our simulation approach has a degree of reliability in predicting extinction risk. While simulations cannot replicate reality with complete accuracy, they provide valuable insights for assessing and comparing the vulnerability of species. It is noteworthy that species within the Cyprinidae family are particularly vulnerable to threats, indicating the non-randomness of the distribution of threatened species in Chinese freshwater fishes and its significant correlation with specific families. The Schizothoracinae subfamily within Cyprinidae exhibits a marked vulnerability, likely linked to their unique distribution in the special aquatic environments of the Tibetan Plateau. The association of Schizothoracinae with the unique environmental conditions of the Tibetan Plateau is correlated with a higher extinction risk, and their distinct life history traits are associated with greater vulnerability to environmental and anthropogenic stressors (although causal links cannot be confirmed with observational data).
We also observe a disproportionately high threat ratio in families with fewer species, such as Acipenseridae, Polyodontidae, Clupeidae, Psilorhynchidae, Gyrinocheilidae, Catostomidae, Plecoglossidae, and Lotidae. This non-random distribution of extinction risk may relate to shared vulnerability traits or characteristics within these families, potentially stemming from evolutionary constraints, natural selection, and environmental pressures. For instance, genetic constraints might reduce some species’ adaptability to environmental changes,31 while natural selection could make others more competitive in specific environments but also more sensitive to environmental fluctuations. Additionally, human activities, including habitat destruction, overfishing, and water pollution, exert extra pressure on these vulnerable species.
This study confirms that extinction risk among Chinese freshwater fish families is non-randomly distributed (chi-square test, p<0.05), characterized by uneven proportions of threatened species across taxa: large families (e.g., Cyprinidae) have high absolute numbers of threatened species, while small families (e.g., Acipenseridae) show extreme proportional threat (100%)—though the latter requires cautious interpretation due to low statistical power. No family exhibited statistically significant overrepresentation of threatened species, but the overall uneven distribution of risk supports the hypothesis of non-random extinction, highlighting the need for family-specific conservation strategies.
It’s important to note that the statistical power of binomial tests is generally lower for families with fewer species.32 This may largely explain why some families with fewer species face higher risks of extinction, yet these risks are not statistically significant. Despite their limited numbers, these species may possess unique biological characteristics and adaptive strategies. Their extinction could lead to a disproportionate loss of fish biodiversity.33 Therefore, conservation efforts should not only focus on families with numerous species but should encompass all species to ensure the diversity and ecological functions of the entire fish community are maintained.
4.2. Extinction Risk Mechanisms in the Schizothoracinae subfamily
This study aims to explore the association between the biological characteristics of the Schizothoracinae subfamily and their extinction risk. Integrating analyses of biological traits like maximum body length, egg diameter, and age of sexual maturity, along with habitat characteristics such as breeding water temperature and habitat altitude, we find no direct correlation between these traits and the extinction risk of Schizothoracinae subfamily species.
Conventional wisdom suggests larger body size as a vulnerability,3 as highlighted by Reynolds et al.,34 who noted the higher extinction risk in large vertebrates. However, our results indicate a possible weakening of this correlation in Schizothoracinae subfamily species, with body length not being the sole factor influencing extinction risk. In freshwater environments, smaller fish may be more dependent on specific habitat conditions and resources. Their shorter bodies, limited movement range, and lower dispersal ability make them more vulnerable to habitat loss or fragmentation. Larger fish, on the other hand, have a wider ecological niche and resource utilization capacity. Under the threat of invasive alien species, they may become more vulnerable targets for predators or human overfishing.33 However, this is not always the case, as illustrated by the larger distribution and higher dispersal ability of the smaller Misgurnus anguillicaudatus compared to the larger Acipenser dabryanus.
Overemphasis on a single trait can overlook the complexity of an ecosystem. The lack of single-trait correlations with extinction risk in Schizothoracinae challenges the generality of traditional trait-based models.3 For plateau-adapted fishes, traits such as body length may be co-adapted with environmental factors (e.g., high-altitude temperature fluctuations) and human disturbances (e.g., habitat fragmentation), resulting in complex, non-linear effects that single-variable analyses cannot capture. This finding suggests that future studies should integrate multi-trait interactions and landscape-level factors. For some larger freshwater fish species, such as the Acipenser baerii, it takes longer to reach sexual maturity, thereby increasing the risk of being caught before breeding.35 Still, these large fish live longer and can reproduce for many years after reaching maturity. Theoretically, larger freshwater fishes might have more opportunities for reproduction, but the reality is often more complex. For instance, Acipenser baerii faces numerous challenges from human activities even after reaching maturity. Human activities such as damming have seriously hindered migration routes, resulting in the loss of spawning grounds.36 Particularly, low river levels in spring lead to mass mortality of eggs and juveniles, exacerbating population declines. Therefore, focusing too much on a single trait may overlook the complexity of the ecosystem. In considering the extinction risk of Schizothoracinae subfamily species, it is crucial to consider both their unique biological traits and the impact of human activities.37
Schizothoracinae subfamily species, as indicator organisms in the Tibetan Plateau, may exhibit unique responses to environmental changes. Our results are different from the traditional view,3 suggesting that Schizothoracinae subfamily has a complex ecological response mechanism to environmental stress. For example, some smaller Schizothoracinae subfamily species might maintain a smaller body size to minimize energy loss, better adapting to temperature fluctuations in high-altitude environments.
This suggests that the response of Schizothoracinae to environmental change is not just a single trait, but involves a more complex ecological response mechanism. Through the study of multidimensional traits, we recognize the potential non-linear and complex interactions among traits like body length, breeding water temperature, and diet in different habitats, affecting their response to external pressures.
We acknowledge a key limitation of this study: the observational, cross-sectional nature of the data prevents us from inferring causal relationships. All correlations reported (e.g., between HFI and conservation status, or between traits and extinction risk) reflect associative patterns rather than direct cause-effect links. For example, the higher prevalence of NT species in high-HFI areas could result from: (1) human activities modifying habitats in ways that favor NT species; (2) NT species inherently occupying habitats that are more likely to be disturbed by humans; or (3) unmeasured confounding factors (e.g., habitat availability, climate suitability). Similarly, the lack of significant trait-extinction risk correlations does not rule out causal effects—instead, it may reflect the complexity of multi-factor interactions or limitations in capturing temporal dynamics. Future studies incorporating long-term temporal data (e.g., tracking HFI and species status changes over decades) or experimental designs (e.g., controlled disturbance experiments) would be necessary to test causal hypotheses about the drivers of extinction risk in Chinese freshwater fishes.
A further limitation is the low statistical power of binomial tests for small families (n≤5 species). While these families show extreme threat proportions (e.g., 100% threatened), we cannot rule out the influence of random variation due to small sample sizes. Future studies with expanded datasets (e.g., including more populations or closely related species) may improve statistical power for these taxa. Additionally, our focus on family-level patterns may mask genus- or species-specific vulnerability, which could be explored in future analyses.
Although the current study has not fully captured the intricate interactions between the various factors, adopting a comprehensive and in-depth research approach is essential to fully understand the multifaceted impacts on extinction risk. This study aims to explore the correlation between the HFI and the conservation status of the Schizothoracinae subfamily. We discovered significant differences in the tolerance of species at different conservation levels to human activities. In interpreting the relationship between the conservation status of Schizothoracinae and changes in human activities, we considered two prevailing hypotheses: the Threat Hypothesis38 and the Refuge Hypothesis.39 The Threat Hypothesis suggests that with intensified land use, the proportion of certain species may increase. This aligns with our observations of near threatened species, which appear to exhibit a degree of adaptability or tolerance to human impacts. This implies that while the increase in human activities poses threats to some species, it may also provide opportunities for adaptation and expansion for others. Conversely, the Refuge Hypothesis posits that certain species may seek refuges in areas with less human activity, leading to a negative correlation between the proportion of endangered species and land-use intensity.40 This is consistent with our findings that endangered species are extremely sensitive to environmental changes and are predominantly found in areas with less human disturbance. Our results support both the Threat and Refuge Hypotheses, revealing the complex impact of human activities on species with different conservation statuses. These findings are crucial for formulating effective species conservation strategies, especially when considering the diverse impacts of human activities on ecosystems.
Our study builds upon previous assessments6,15,17 by integrating phylogenetic uniqueness, statistical validation of extinction risk bias, and the effects of human disturbance. The identification of high-PII species, non-random threat distribution, and divergent species responses to HFI provides a more holistic framework for freshwater fish conservation in China. The non-significant trait–risk correlations highlight the need to rethink extinction risk drivers for extremophile groups, offering new directions for future research on plateau aquatic ecosystems. In conclusion, while our study makes valuable contributions to the understanding of species extinction risk, the complexity of ecological systems necessitates a cautious interpretation of these findings. We recommend adopting a comprehensive approach in future biodiversity research, encompassing a broader spectrum of variables and more nuanced analytical methods, to fully unravel the complexity of factors influencing species extinction risk.
Acknowledgments
This study is financially supported by National Natural Science Foundation of China (grant no. 32273140).
Authors’ Contribution
Methodology: Dan Song (Equal), Qiqun Cheng (Equal). Formal Analysis: Dan Song (Equal), Qiqun Cheng (Equal). Investigation: Dan Song (Equal), Qiqun Cheng (Equal). Writing – original draft: Dan Song (Lead). Writing – review & editing: Dan Song (Lead), Qiqun Cheng (Lead), Sumaiye Mieradili (Equal), Di Peng (Equal), Biyuan Liu (Equal). Resources: Dan Song (Equal), Qiqun Cheng (Equal). Conceptualization: Qiqun Cheng (Lead). Funding acquisition: Qiqun Cheng (Lead). Supervision: Qiqun Cheng (Lead).
Competing of Interest – COPE
The authors declare that they have no known competing financial interests.
Ethical Conduct Approval – IACUC
This study exclusively utilized publicly accessible data, such as China’s Red List of Biodiversity,12 FishBase,25 and Taiwan Fish Database.26 No live animals, animal tissues, or plants were involved in this study. Because the dataset is publicly available, no Institutional Animal Care and Use Committee (IACUC) approval is required.
Informed Consent Statement
All authors and institutions have confirmed this manuscript for publication.
Data Availability Statement
All are available upon reasonable request.