1. INTRODUCTION
Pacific white shrimp (Litopenaeus vannamei) can be farmed at high density due to its fast growth and strong environmental adaptability.1 In 2023, its output in China reached 2.24 million tons.2 The number of greenhouses in China reached 600,000, with an annual output of 612,000 tons of L. vannamei, accounting for 27.3% of the total shrimp production.2 With the increasing demand for aquatic products, aquaculture has tended towards more high-density and large-scale farming models.3,4 In the greenhouses shrimp farming model, aquatic animals are increasingly threatened by diseases. However, the extensive use of antibiotics and chemical agents has caused serious impacts on the quality of aquatic products and the water environment. Therefore, the application of safe, environmentally friendly, and healthy agents is the trend of the future development of the aquatic industry.
In China, the most promising immune enhancer for is compound Chinese herbals (Chs). Chs contains many active ingredients, such as organic acids, polysaccharides, alkaloids and flavonoids, etc. These active ingredients are closely related to the immune function, metabolism and growth performance of aquatic animals.5 Therefore, Chs has also attracted the attention of a large number of researchers.6,7 The application of Chs in aquaculture has a long history and its sources are extensive, mainly coming from the roots, stems, leaves, flowers, fruits, and other parts of plants. After being processed by scientific methods to remove harmful components and through long-term practice, the application of Chs is safe and reliable.8 Studies have shown that Chs increased the appetite of aquatic animals, promoted metabolism, enhanced immunity, reduced the occurrence of diseases, and also promoted growth.9,10 It is reported that the application of dietary Chs additives in red claw crayfish (Cherax quadricarinatus),11 grass carp (Ctenopharyngodon idellus),12 juvenile Japanese seabass (Lateolabrax japonicus)13 and Nile tilapia (Oreochromis niloticus)14 reduced the occurrence of diseases during the breeding process, thereby achieving better growth performance.
Chs is a drug composed of two or more Ch in a certain proportion. Compared with a single Ch, Chs can exert the compatibility effect among different drug components and achieve better results.15 Although many studies have shown that Chs are beneficial to the growth of aquatic animals, most were laboratory experiments, and few have investigated the effects of Chs in actual production processes. Does the application of Chs in the feed for L. vannamei show different effects in the laboratory and in production? In this context, the present experiment evaluated feeds with and without Chs that were administered to L. vannamei reared in greenhouse conditions to assess the effects of dietary Chs on the hepatopancreas (HP) and intestinal health, antioxidation, and non-specific immunity of L. vannamei. Overall objective of this work was to explore the impact of Chs on L. vannamei to promote the green and healthy development of aquaculture.
2. METHODS
2.1. EXPERIMENTAL DIETS AND DESIGN
Two types of commercial feed were used in the experiment, namely the control feed (CF) and the Chs feed (ChF). The ChF was formulated by adding 2% compound Chs to the CF and reducing the wheat flour content, forming two groups of isonitrogenous and isolipid feeds. The composition of the compound Chs are artemisia annua (Artemisia annua L.), astragalus (Astragalus membranaceus), isatis indigotica (Isatis indigotica Fort.), polygonatum (Polygonatum sibiricum Redouté) and licorice (Glycyrrhiza uralensis Fisch.), and the ratio of the five herbs is 1:1:1:1:2. These Chinese herbals were purchased from the specialized market for medicinal herbs in Bozhou, Anhui Province. Fish meal, corn protein meal, meat and bone meal, soybean meal, peanut meal, brewer’s yeast, krill meal, and squid paste are the main protein sources in feed. Fish oil was added to the feed to meet the fat requirements of L. vannamei. The use of corn protein meal, soybean meal, and peanut meal has met the carbohydrate requirements. The moisture content of both feeds is less than 12%, and their protein and fat content is 42% and 6%, respectively. Both types of commercial feed are provided by Jiangsu Changshou Group Nanshan Feed Co., LTD.
The experiment was divided into two groups, with four parallels in each group (n = 4). Each greenhouse is considered a separate parallel, and there is a total of 8 such greenhouses. Two types of feed were fed to L. vannamei farmed in greenhouses for 12 weeks. Through sampling in the middle and later stages, various indicators were detected to determine the effects of Chs on the HP and intestinal health, antioxidation and non-specific immunity of L. vannamei. The two samplings were respectively the mid-stage of CF and ChF groups (CF-M, ChF-M), the late-stage of CF and ChF group (CF-L, ChF-L), which formed the above four comparisons.
2.2. EXPERIMENTAL SHRIMP AND FEEDING MANAGEMENT
The feeding trial experiment was conducted in commercial outdoor rectangular greenhouses pools (50.0 m × 10.0 m) in Nantong China. All the greenhouses are of the same size. The middle area is deeper (1.2 m), while the surrounding area is shallower (0.7 m). The ratio of the pond’s slope to its depth is approximately 1:1. A cement slab about 30 cm wide is set up in the middle of the pond as a passage, which can be used for passage and viewing. Above the pond, there is an aquaculture shed about 1.8 meters high, constructed with arc-shaped steel, with plastic film laid on the outside for insulation. The greenhouses were equipped with oxygenators to prevent hypoxia during the breeding process. One nano-oxygenation tray was placed every 2 meters, and each nano-oxygenation tray was wrapped with 5 meters of nanotubes. The nano-oxygenation tray is about 10 centimeters higher than the bottom of the pool. Before stocking, 1,200 kilograms of bleaching powder per hectare was used for pond cleaning and disinfection. After the water quality is suitable, 50,000 shrimp fry were released into each greenhouse.
During the rearing period, the daily feeding amount was 3% to 10% of the shrimp’s body weight. The daily feeding amount was divided into four portions (06:00, 12:00, 17:00 and 22:00). Specific feeding amounts were adjusted in a timely manner based on the feeding situation and weather conditions, ensuring that the feeding amounts for each parallel were basically the same, and guaranteeing that the feed was consumed within 2 hours after feeding. During the feeding trial, water temperature, salinity, dissolved oxygen, ammonia, and pH were 25–32℃, 1.0‰~3.0‰, >6.0 mgL-1, <0.05 mgL-1, and 7.5 - 8.4, respectively. The water quality parameters were tested by ProQuatro/1020 (YSI, American). The feeding experiment lasted 12 weeks.
2.3. SAMPLING
During the mid-term and late-stage sampling, 20 shrimps were randomly collected from each pool. After anesthetizing the shrimps in ice water, 4 shrimps were randomly selected in each parallel. 0.5 mL of hemolymph was drawn from the venous sinus using a 1.0 mL sterile syringe and placed in a 1.5 mL centrifuge tube pre-added with 0.5 mL of anticoagulant. Centrifuged for 20 minutes at 12,000 r min-1 and 4°C. The supernatant was collected and stored at -80 ℃ for later use. After taking hemolymph, the cephalothorax was opened, and the HP were taken and respectively placed in 5 mL centrifuge tubes for the determination of digestive enzyme activity. Another 4 shrimp were sampled, the HP were collected respectively and stored in 10 mL centrifuge tubes pre-filled with 7 mL of Bouin fixative for the preparation of HP tissue sections. The intestines of the above 4 shrimps were collected and placed in 5 mL centrifuge tubes pre-filled with 3 mL of Bouin fixative for the preparation of intestinal tissue sections. Four additional shrimps were sampled from each parallel. Their body surface was wiped with 75% alcohol for sterilization. Under aseptic conditions, gill tissues were collected for the determination of immune-related gene expression levels. The mid-intestinal were sampled and placed in a 1.0 mL centrifuge tube for microbial study using 16S rRNA sequencing.
2.4. SERUM BIOCHEMICAL PARAMETERS
Acid phosphatase (ACP), alkaline phosphatase (AKP), and total antioxidant capacity (T-AOC) were determined by the colorimetry method. Lysozyme (LZM) was determined by turbidimetry methods. Catalase (CAT) was determined by the complex method. Total superoxide dismutase (T-SOD) was determined by the xanthine oxidase method. Malondialdehyde (MDA) was determined by the thiobarbituric acid method. Glutathione peroxidase (GSH-PX) was determined by the nitrobenzoic acid method. All measurements were conducted according to the instructions for test kits produced by Nanjing Jiancheng Bioengineering Institute (Nanjing, China).
2.5. HP DIGESTIVE ENZYME ACTIVITY
Frozen HP was mixed with 9 times saline (W:V = 1:9), then homogenized and centrifuged at 4 ℃ for 15 min (3000 r min-1). The supernatant was collected for the determination of the digestive enzyme activity. Protease activity was measured where one unit was defined as 1 μg tyrosine released per minute by hydrolyzing casein at pH value of 7.2 (1 mg tissue protein). One unit of amylase activity was defined as the amount of enzyme that catalyzes the reaction between 1 g of tissue protein and the substrate, consuming 1 µmol of the substrate at 37 ℃ in 1 min. One unit of amylase activity was defined as 10 mg starch hydrolyzed in the reaction per 30 min at 37 ℃ (1 mg tissue protein), and the test-kit was provided by Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Tissue protein content was determined by Coomassie brilliant blue method.
2.6. HP AND INTESTINAL HISTOLOGY
During the sectioning process, the mid-intestine and HP were dehydrated with ethanol, transparented with xylene, embedded in paraffin, sectioned (with a thickness of 5μm) (Leikn RM2235 sectioning machine, Germany), stained with HE, and sealed to produce permanently preserved sections. Under a light microscope, the morphological characteristics of intestinal villus were observed and photographed (Nikon YS100 microphotography system), and the height, width and muscular layer thickness of intestinal epithelial folds were measured with a micrometer.
2.7. EXPRESSION LEVELS OF IMMUNE-RELATED GENES IN GILL TISSUES
The specific primer pairs for the β-actin gene and other genes (Table 1) were designed based on the relevant cDNA sequences of L. vannamei in the National Center for Biotechnology Information (NCBI) database.
Total RNA was extracted using Trizol reagent. The concentration and purity of total RNA were detected by ultraviolet spectrophotometer and agarose gel electrophoresis. Total RNA was reverse transcribed into cDNA using TaKaRa reverse transcription kit (PrimeScriptTM RT). The fluorescence quantitative PCR reaction procedure is as follows: 3 min of pre-denaturation at 95℃, 10 seconds of denaturation at 95℃, 10 seconds of annealing at 60℃, 30 seconds of extension at 72℃, for a total of 39 PCR cycles, and finally a melting curve detection is performed. Taking the gene expression level of the gill tissue of the CF-M group of L. vannamei as the reference, with β-actin as the internal reference gene, the expression levels of immune-related genes in the gill tissues of each group were calculated using the 2-ΔΔCt method.
2.8. INTESTINAL MICROBIAL STUDY
The sequencing experiment was entrusted to Shanghai Majorbio Bio-pharm Technology Co., Ltd. (China) for next-generation sequencing (NGS) library preparation and Illumina Miseq sequencing. The specific method is as follows:
Total microbial genomic DNA was extracted from the intestine using the FastPure Stool DNA Isolation Kit (MJYH, Shanghai, China) according to the manufacturer’s instructions. The quality and concentration of DNA were determined by 1.0% agarose gel electrophoresis and a NanoDrop® ND-2000 spectrophotometer (Thermo Scientific Inc., USA) and kept at -80 ℃ prior to further use. The hypervariable region V3-V4 of the bacterial 16S rRNA gene was amplified with primer pairs 338F (5’-ACTCCTACGGGAGGCAGCAG-3’) and 806R(5’-GGACTACHVGGGTWTCTAAT-3’) by a T100 Thermal Cycler (BIO-RAD, USA). The PCR reaction mixture included 4 μL 5 × Fast Pfu buffer, 2 μL 2.5 mM dNTPs, 0.8 μL each primer (5 μM), 0.4 μL Fast Pfu polymerase, 10 ng of template DNA, and ddH2O to a final volume of 20 µL. PCR amplification cycling conditions were as follows: initial denaturation at 95 ℃ for 3 min, followed by 27 cycles of denaturing at 95 ℃ for 30 s, annealing at 55 ℃ for 30 s and extension at 72 ℃for 45 s, and single extension at 72 ℃ for 10 min, and end at 4 ℃. All samples were amplified in triplicate. The PCR product was extracted from 2% agarose gel and purified. Then quantified using Synergy HTX (Biotek, USA).
Purified amplicons were pooled in equimolar amounts and paired-end sequenced on an Illumina NextSeq 2000 PE300 platform (Illumina, San Diego,USA) according to the standard protocols by Majorbio Bio-Pharm Technology Co. Ltd. (Shanghai, China). After demultiplexing, the resulting sequences were quality-filtered with fastp (v0.19.6) and merged with FLASH (v1.2.11). Then the high-quality sequences were denoised using the DADA2 plugin in the Qiime2 (version 2020.2) pipeline with recommended parameters, which obtains single-nucleotide resolution based on error profiles within samples. Taxonomic assignment of ASVs was performed using the Naive bayes consensus taxonomy classifier implemented in Qiime2 and the SILVA 16S rRNA database (v138.2). Bioinformatic analysis of the gut microbiota was carried out using the Majorbio Cloud platform (https://cloud.majorbio.com).
2.9. STATISTICAL ANALYSIS
For statistical analysis, the greenhouse was considered the experimental unit (n=4). The experimental data are presented as mean ± standard deviation. All data were analyzed using SPSS 22.0 statistical software. A one-way analysis of variance (ANOVA) and Turkey’s multiple range tests were used to determine the statistical significance among groups. Statistical significance was determined at P < 0.05.
3. RESULT
3.1. SERUM BIOCHEMICAL INDICES
As shown in Table 2, the serum ACP activity of the ChF-M and ChF-L groups was significantly higher than that of the CF-M and CF-L groups, and the MDA content was significantly lower than that of the CF-M and CF-L groups (P < 0.05). Serum LZM and T-AOC activities of ChF-M group were significantly higher than those of the CF-M group (P < 0.05). Serum GSH-Px activity of ChF-L group was significantly higher than that of the CF-L group (P < 0.05). There was no significant difference in ALP and CAT activities among all groups (P > 0.05).
3.2. HP DIGESTIVE ENZYME ACTIVITY
As shown in Table 3, HP protease and amylase activities of ChF-M and ChF-L groups were significantly higher than those of the CF-M and CF-L groups (P < 0.05). There was no significant difference in HP lipase activity among all groups (P > 0.05).
3.3. EXPRESSION OF IMMUNE-RELATED GENES IN GILL TISSUE
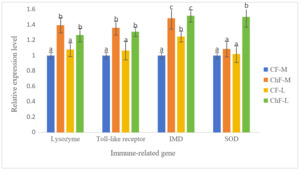
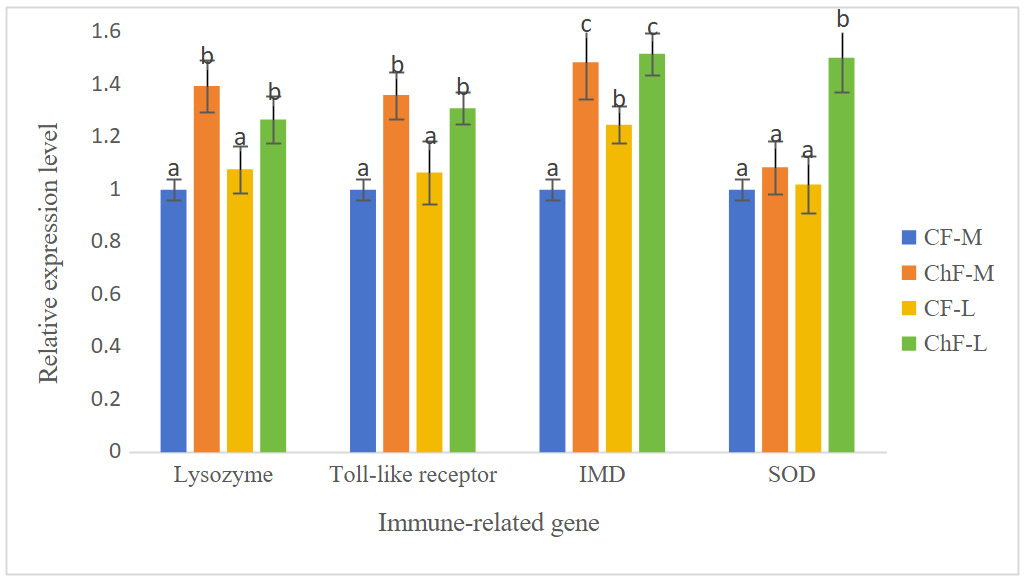
As shown in Figure 1, the expression of the gill lysozyme, toll-like receptor and IMD genes in ChF-M and ChF-L groups were significantly higher than those of the CF-M and CF-L groups (P < 0.05). The expression of the gill SOD gene in ChF-M group was significantly higher than that of other three groups (P < 0.05).
3.4. HP AND INTESTINAL HISTOLOGY
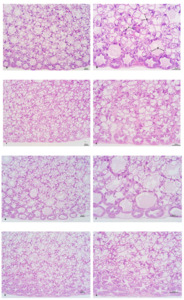
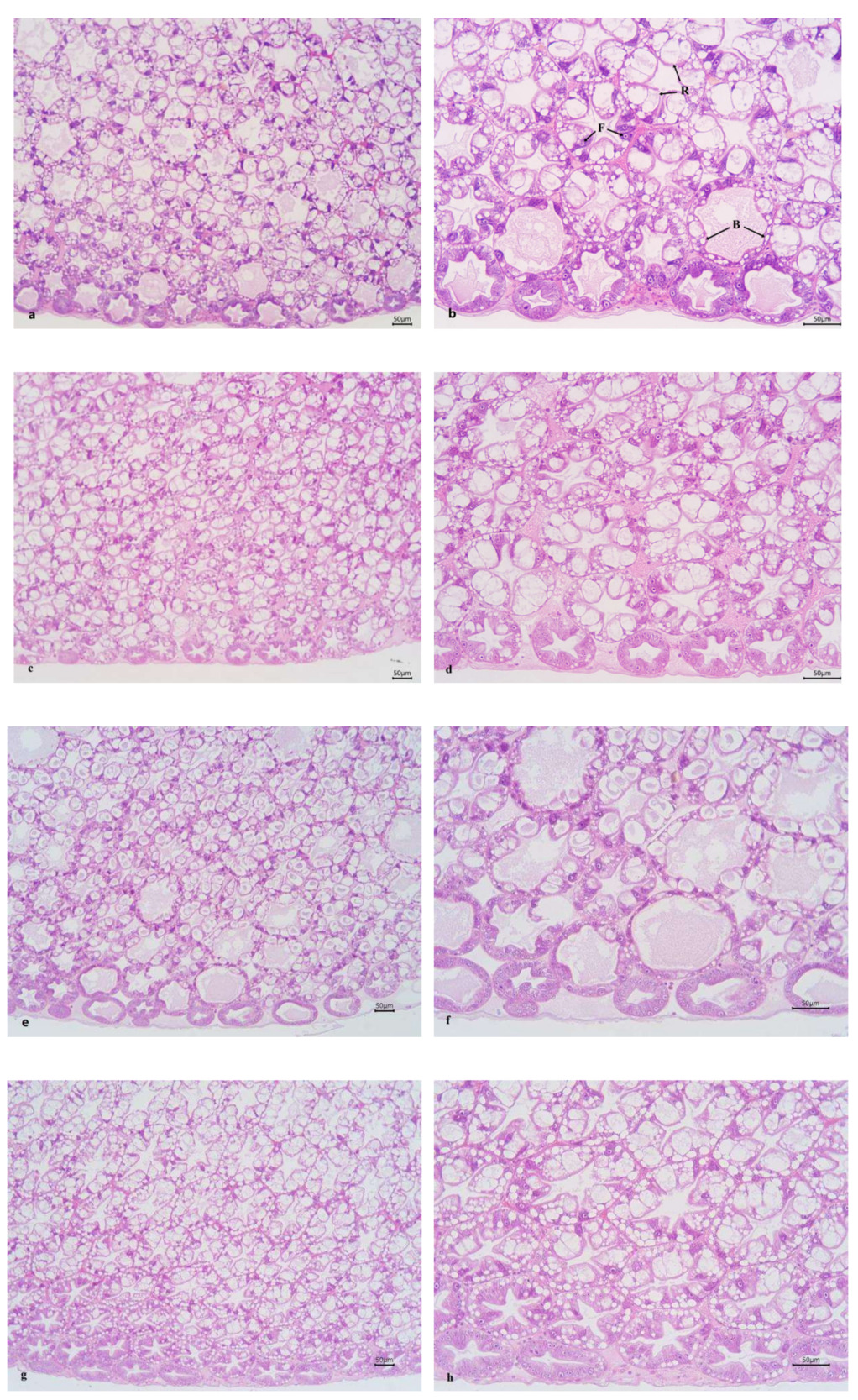
The intercellular junctions of the HP tubular epithelial cells in the ChF-M and ChF-L groups were well-organized. The HP tubules in these groups had a star-shaped and polygonal structure, with clear morphology, moderate lumen size and epithelial thickness. In the CF-M and CF-L groups, the star-shaped and polygonal structures of some HP tubules disappeared, the lumen became larger, the epithelium became thinner, and B- and R-cells were compressed into oval shapes (Figure 2).
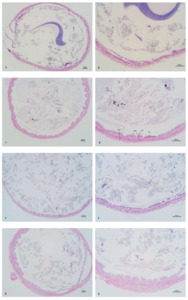
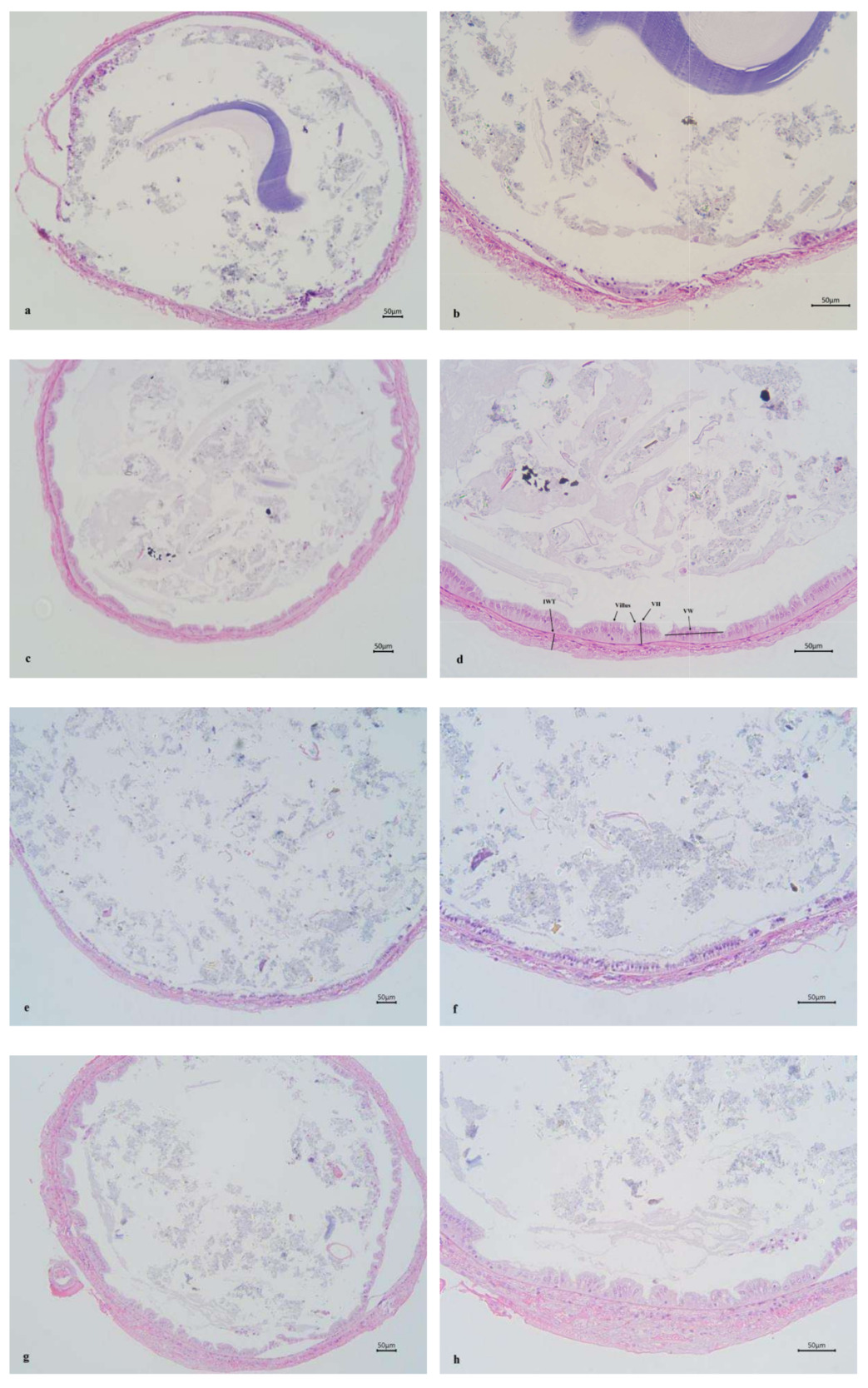
Figure 3 shows the mid-intestinal morphology of L. vannamei fed different diets at different times. The ChF-M and ChF-L groups had well-arranged and closely-packed intestinal villus. However, the CF-M and CF-L groups had significantly reduced folds height and intestinal wall thickness (P < 0.05), and even only a single layer of columnar cells was present, with no obvious intestinal folds.
Mid-intestinal villus height, width and wall thickness of ChF-M and ChF-L groups all significantly higher than those of the CF-M and CF-L groups (P < 0.05) (Table 4).
3.5. MICROBIAL STUDY IN INTESTINE USING 16S rRNA SEQUENCING
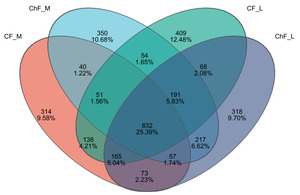
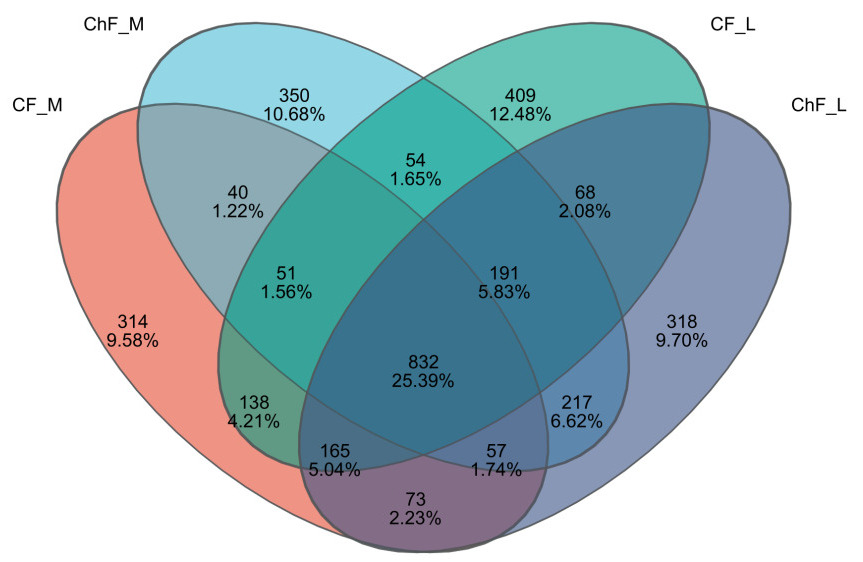
In this study, a total of 1092637 valid sequences were obtained with sequence lengths ranging from 200 to 476 bp. After cluster analysis of the obtained sequences, five groups obtained 1670, 1792, 1908 and 1921 representative bacterial communities OTUs, respectively. The OTUs shared by the five treatment groups are 823, while the unique OTUs are 314, 350, 409 and 318, respectively (Figure 4).
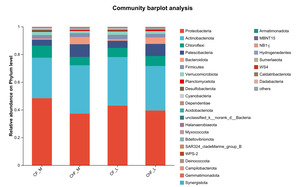
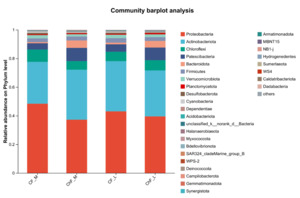
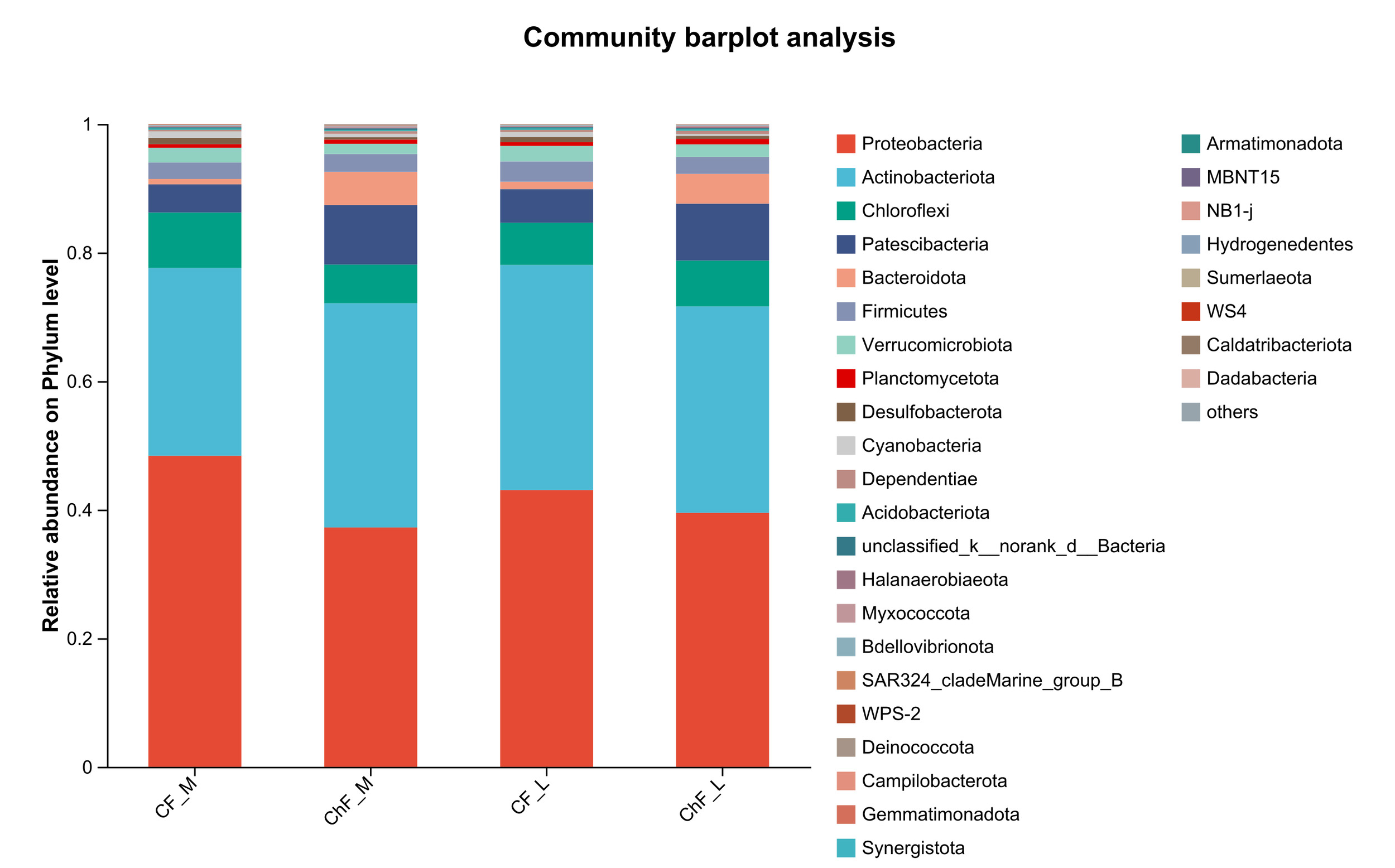
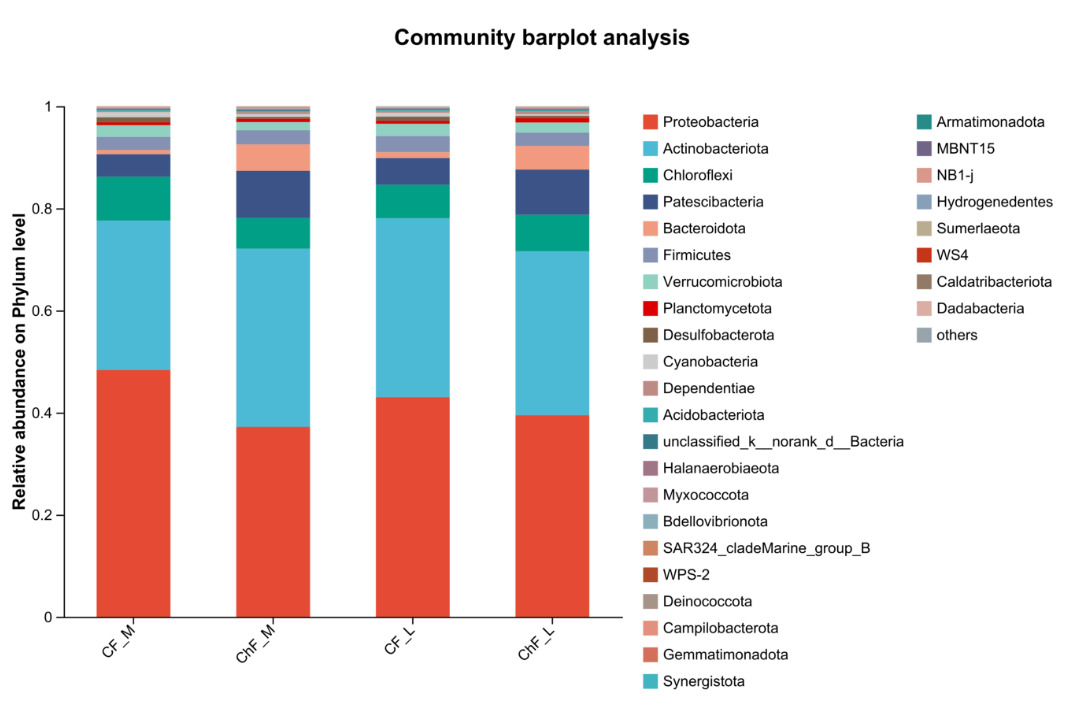
In Figure 5, the composition of intestinal microflora at the phylum level included Proteobacteria, Actinobacteriota, Chloroflexi and Patescibacteria, accounting for 93.17%, 95.34%, 94.17% and 94.86% abundance of the intestinal microbiota, respectively. The rest mainly include Verrucomicrobiota, Planctomycetota, Desulfobacterota, Cyanobacteria and Dependentiae. There was no significant difference in the composition and abundance of microbiota among the treatment groups (as there was no difference, no bar plot was generated).
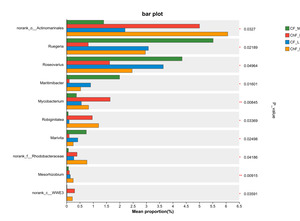
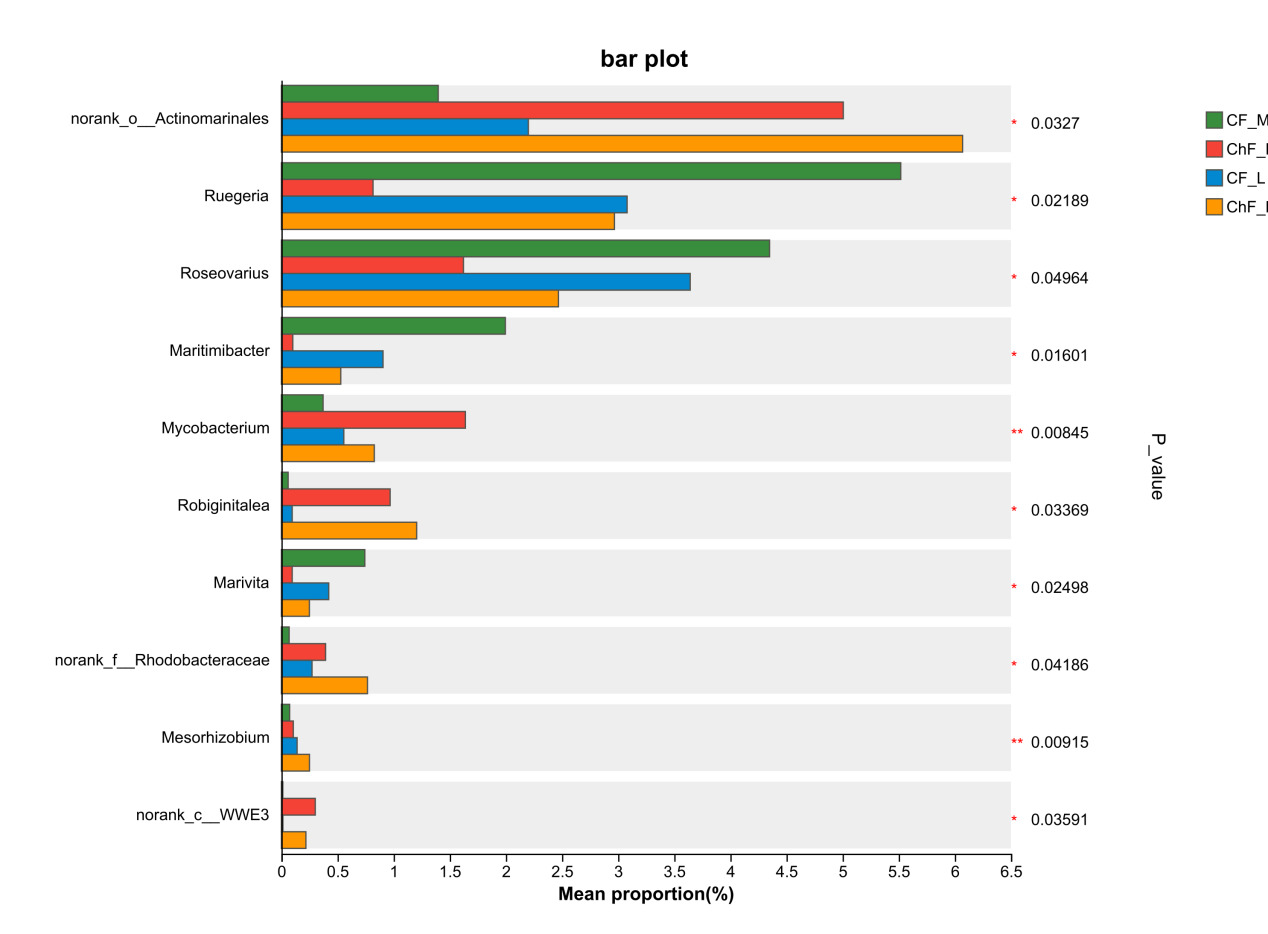
At the genus level, the dominant intestinal microflora included unclassified_f__Rhodobacteraceae, norank_f__Intrasporangiacea, unclassified_o__Micrococcales, norank_f__JG30-KF-CM45, norank_o__Actinomarinales, norank_o__Saccharimonadales and Ruegeria (Figure 6). There were significant differences in the abundance of norank_o__Actinomarinales, Ruegeria, Roseovarius, Maritimibacter, Robiginitalea, Marivita, norank_f__Rhodobacteraceae, Mesorhizobium and norank_c__WWE3 (P < 0.05) (Figure 7).
4. DISCUSSION
4.1. HP HISTOLOGY AND DIGESTIVE ENZYME ACTIVITY
HP is directly involved in the synthesis and secretion of digestive enzymes, and subsequently participates in absorption, nutrient assimilation, and waste excretion.16,17 In addition, HP also stores important nutrients such as lipids, glycogen, and other organic and inorganic compounds.18 The morphology of the HP reflects the quality of the external environmental conditions and shows great plasticity among different species.19,20 Meanwhile, HP is also the largest digestive organ in crustaceans and can be used to analyze the organism’s nutritional status.21 The hepatic tubules are regarded as the morphological and functional units of HP.22,23 They are composed internally of digestive epithelial cells, including E- (embryonic), F- (fibrous), B- (blasenzellen) and R- (restzellen) cells.24 When analyzed through histological and histochemical methods, each cell type exhibits distinct characteristics.20 Studies have reported that a compound Chs (Artemisia capillaris, Gardenia jasminoides, Scutellaria baicalensis, Reynoutria japonica and Uncaria rhynchophylla) has hepatoprotective effects and protected the liver of juvenile large yellow croaker (Larimichthys crocea).25 Zhou et al.26 also made similar findings in their research on Jian carp (Cyprinus carpio). Lu et al.27 discovered that dietary Eucommia ulmoides leaf extract effectively improved the HP morphology and enhanced the absorption function of various nutrients in red claw crayfish. In this experiment, the intercellular junctions of the HP tubular epithelial cells in the ChF-M and ChF-L groups were well-organized, and the HP tubules presented a star-shaped polygonal structure with clear morphology, moderate lumen size and epithelial thickness. However, in the CF-M and CF-L groups, the star-shaped polygonal structure of some HP tubules disappeared, the lumen became larger, the epithelium became thinner, B- and R-cells were compressed into oval shapes. Thus, dietary Chs effectively improved the morphological structure of HP tubular cells in L. vannamei.
Huang et al.28 found that dietary compounds Astragalus membranaceus, Eucommia ulmoides, and Artemisia argyi increased the intestinal trypsin activity of European eel (Anguilla anguilla). When Lycium barbarum polysaccharides, Astragalus, and Codonopsis pilosula, oregano essential oil were respectively added to the diets of Chinese mitten crab (Eriocheir sinensis),29 L. vannamei,30 and carp31,32 respectively, it was found that all improved the digestive enzyme activity of the farmed animals. In this experiment, we found that the activities of HP protease and amylase of the ChF-M and ChF-L groups were significantly higher than those of the CF-M and CF-L groups. This might be due to the change in the morphology of B-cell in the CF-M and CF-L groups (as the HP tissue structure has been damaged), which led to a decrease in the secretion of digestive enzymes compared to the ChF-M and ChF-L groups. Studies have shown that the morphological changes in HP are related to the changes in digestive enzyme activity.4 The exocrine function of the pancreas is the main site for the synthesis and secretion of trypsin, lipase and amylase in aquatic animals.32 Among them, B cells are the main site of synthesis of digestive enzymes.33 Changes in the morphology of B-cells may affect the activity of digestive enzymes in HP. Based on the experimental results, the HP tubules in the group with Chs added showed better morphological and cellular structure and higher digestive enzyme activity. These results indicated that dietary Chs promotes the absorption and utilization of nutrients, thereby favorably impacting L. vannamei.
4.2. ANTIOXIDATION AND NON-SPECIFIC IMMUNITY
The antioxidant defense mechanisms, including T-AOC, CAT and GSH-PX, can protect host cells from oxidative damage.34 SOD can eliminate superoxide anions in the body and maintain the redox balance.35 The primary function of CAT is to eliminate excessive hydrogen peroxide and maintain the cellular redox balance.36 GSH-Px can catalyze the reduction of hydrogen peroxide to water or to the corresponding alcohol. This reduction reaction uses GSH as the reducing agent, removing hydrogen peroxide and other peroxides from the body to maintain normal physiological functions.28 Antioxidant defense has been extensively studied in crustaceans to measure antioxidant capacity and for disease monitoring. However, oxidative stress can produce lipid peroxides, including MDA. MDA is the main oxidative product of polyunsaturated fatty acids during lipid peroxidation. It can damage the structure of lipid membranes, thereby significantly impacting normal cellular functions.37 Studies have shown that dietary active components (Astragalus polysaccharide, chlorogenic acid, and berberine) significantly enhanced the antioxidant capacity of red claw crayfish.11 Zhou et al.38 conducted a study on juvenile largemouth bass (Micropterus salmoides) and found that dietary herbal ingredients (honeysuckle, dandelion, scutellaria, and astragalus) increased the liver SOD, GSH-PX, T-AOC, and CAT activities, and reduced the content of MDA. This indicates that these herbs effectively enhanced the antioxidant capacity of juvenile largemouth bass and reduced the damage caused by oxidative stress to the body. At present, numerous studies have shown that dietary Chs or its active components promote the antioxidant system in aquatic animals.28 In this experiment, dietary Chs significantly increased serum SOD and GSH activities and gill SOD gene expression levels, and significantly decreased serum MAD content, results that were generally consistent with those of previous studies.
Crustaceans can only rely on their innate immune system, which lacks specific antibodies and immune memory, to defend against the invasion of foreign substances.39 Therefore, the non-specific immune system is particularly important for crustaceans. ACP, ALP, and LZM are important non-specific immune enzymes in the body fluids of crustaceans. In an acidic environment, ACP exerts an immune function by recognizing and catalyzing the hydrolysis of phosphate bonds of foreign substances to degrade or destroy pathogens.40 ALP is an important metabolic regulatory enzyme that, together with other hydrolases, forms an important detoxification system.41 LZM hydrolyzes the bacterial cell wall, leading to bacterial death, and can also induce the synthesis of other immune-related factors.42 Previous studies have shown that Chs active components (astragalus polysaccharides, chlorogenic acid, and berberine) significantly enhanced the immune function and antioxidant capacity of red claw crayfish.11 Wang et al.43 and Yin et al.44 made similar findings in rainbow trout (Oncorhynchus mykiss) and Nile tilapia, respectively. In this experiment, we found that dietary Chs significantly increased serum ACP and LZM activities and gill lysozyme gene expression, consistent with the results of the aforementioned studies. This might be attributed to the components of astragalus and licorice. Astragalus has a wide range of biochemical and pharmacological activities,45 which can enhance immune regulation, antioxidant capabilities.46 The roots of licorice contain glycyrrhizin, which can improve immune regulation, antioxidant, antiviral, and anti-inflammatory properties.47 In the studies on Chinese mitten crab,48 and common carp (Cyprinus carpio),49 it was found that astragalus or licorice effectively enhanced the immunity of the farmed animals, which is consistent with the results of this experiment. These results indicate that dietary Chs enhanced the immune capacity of L. vannamei, enabling them to better resist external viral invasion and increase their survival rate.
4.3. INTESTINAL HEALTH
Intestine is the largest interface between aquatic animals and their external environment. It not only performs the functions of digestion and absorption but also acts as a barrier to prevent harmful substances from entering the bloodstream.50 Therefore, the integrity and stability of the structure and function of the intestine are of vital importance for its nutritional digestion and absorption. The parameters of the intestinal morphology of aquatic animals mainly include villus width, villus height and muscle layer thickness, which are directly related to the growth of aquatic animals. The villus height and muscle layer thickness are important physical indicators reflecting the intestinal absorption efficiency.51 The villus height indicates the area where the villi interact with nutrients, and the muscle layer thickness directly reflects the intensity of intestinal contraction. Studies have reported that herbs mixture (Artemisia capillaris, Gardenia jasminoides, Scutellaria baicalensis, Reynoutria japonica and Uncaria rhynchophylla) enhanced the structural integrity of the intestines in large yellow croaker.25 Similar findings were also observed in studies of hybrid grouper (Epinephelus fuscoguttatus ♀ × Epinephelus lanceolatus ♂).52 In this study, the intestinal villus of the shrimp in the ChF-M and ChF-L groups were arranged orderly and closely. However, the villus height of and the intestinal wall thickness of CF-M and CF-L groups have significantly decreased, and only a single layer of columnar cells was present, with no obvious intestinal villus. Thus, the addition of the compound Chs effectively improved the intestinal tissue structure of L. vannamei.
Intestinal microbiota refers to the total community of microorganisms in the intestinal tract of an organism. All the functions of the intestine are achieved through the metabolism of bacteria within it. The intestinal microbiota benefits the host by improving the immune response, nutrient absorption, disease defense, and maintaining the dynamic balance within the body, and is closely related to the host’s health.53 In recent years, there have been numerous reports on the intestinal microbiota of L. vannamei, and it has attracted the attention of many researchers.54,55 Based on the studies by Duan et al.,55 the dominant bacterial phyla in the intestines of the L. vannamei at the phylum level are Proteobacteria, Firmicutes, Bacteroidetes and Actinobacteria. In this study, the dominant bacterial phyla in each treatment group of L. vannamei were Proteobacteria, Actinobacteriota, Chloroflexi and Patescibacteria, and they had similar compositions. However, in this study, the abundances of the Chloroflexi and Firmicutes phyla were higher, while the abundance of the Proteobacteria phylum was lower. This may due to the abundance of intestinal microbiota can change due to factors such as the breeding environment and diet. Studies have reported that dietary herbal additives affected the relative abundance of intestinal flora in farmed animals.56,57 Su et al.58 discovered that dietary herbs significantly increased the abundance of beneficial bacteria such as Pseudomonas fluorescens. In this experiment, we found that the abundances of Actinomycetes and Rhodobacteraceae in the ChF-M and ChF-L groups significantly increased. Actinomycetes is a type of Gram-positive bacteria that are widely distributed in soil, air, water bodies, and the intestines of animals. They are extremely important microorganisms. The Rhodobacteraceae belongs to the α-proteobacteria family. It is one of the probiotics widely present in aquaculture, playing a significant role in improving water quality, promoting the health of aquatic animals, and enhancing the efficiency of aquaculture.59 Therefore, it can be seen that dietary Chs effectively increased the relative abundance of beneficial bacterial species in the intestines, thereby having a beneficial effect on L. vannamei.
5. CONCLUSSIONS
In the present study, dietary compound Chs (Artemisia annua L., Astragalus membranaceus, Isatis indigotica Fort., Polygonatum sibiricum Redout and Glycyrrhiza uralensis Fisch.) effectively improved the HP and intestinal health, antioxidation and non-specific immunity of L. vannamei in greenhouses shrimp farming model.
ACKNOWLEDGMENTS
This work was financially supported by Jiangsu Agri-animal Husbandry Vocational College school level research project (NSF2025ZR08) and Jiangsu Changshou Group Nan Shan Feed Co., Ltd. (S20240005).
AUTHORS’ CONTRIBUTION
Methodology: Jianguo Wang (Equal), Chunyan Zhang (Equal). Formal Analysis: Jianguo Wang (Equal), Jiaxin Li (Equal), Qiuyang Ni (Equal), Ping Gu (Equal), Xuesen Chai (Equal). Investigation: Jianguo Wang (Equal), Chunyan Zhang (Equal). Writing – review & editing: Jianguo Wang (Equal), Chunyan Zhang (Equal). Project administration: Jianguo Wang (Equal), Chunyan Zhang (Equal), Ping Gu (Equal), Xuesen Chai (Equal), Wenxiang Yao (Equal). Conceptualization: Jianguo Wang (Equal), Chunyan Zhang (Equal). Funding acquisition: Jianguo Wang (Equal), Chunyan Zhang (Equal), Wenxiang Yao (Equal). Writing – original draft: Jiaxin Li (Equal), Qiuyang Ni (Equal).
ANIMAL ETHICS STATEMENT
All animal care and use procedures were approved by the Institutional Animal Care and Use Committee of Jiangsu Agri-animal Husbandry Vocational College (Permit number: jsahvc-2023–17), and all authors clearly indicated that such guidelines have been followed.
DECLARATION OF COMPETING INTEREST
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.
DATA AVAILABILITY
Data will be made available on request.