Introduction
Carbohydrates are an economical and readily available source of energy in aquaculture. Adequate dietary carbohydrate intake can spare lipids and proteins in fish by reducing the catabolism of other nutrients.1,2 Furthermore, improving carbohydrate utilization is essential for the long-term sustainability of aquaculture.3 However, fish are less efficient at utilizing carbohydrates compared to mammals, as they require more time to clear excess glucose from the bloodstream,4,5 and have not developed effective internal mechanisms to regulate hyperglycemia over the course of their evolutionary history.6 Therefore, exploring the use of exogenous additives to enhance carbohydrate utilization in fish represents a promising strategy to improve aquaculture efficiency.
Tea (Camellia sinensis) is one of the most widely consumed beverages worldwide, celebrated not only for its refreshing taste but also for its numerous health benefits.7,8 Among the bioactive compounds in tea, tea polysaccharides (TPS) have garnered significant attention due to their diverse therapeutic effects, including antitumor, antioxidant, hepatoprotective, and antidiabetic properties.9–11 Notably, TPS exhibits low toxicity and is recognized as safe for oral consumption, positioning it as a promising natural food additive. In contrast to other carbohydrate-utilization enhancers in fish feeds, such as probiotics and synthetic regulators, TPS presents distinct advantages. Derived from a natural source, TPS aligns with the increasing emphasis on sustainable and eco-friendly aquaculture practices.7,8 Moreover, its high biocompatibility and favorable safety profile offer a potentially safer alternative to single-target or synthetic additives, which may carry risks of side effects.12 Given these attributes, TPS-supplemented aquafeeds hold considerable promise. This is further supported by its successful application as a dietary additive in poultry,13,14livestock,15and fish.16,17 Previous studies in aquatic species indicate that dietary TPS can improve growth performance,18 boost immune responses,19,20and elevate digestive enzyme activities.21 Nevertheless, despite these documented benefits, little attention has been paid to the specific role of TPS in modulating carbohydrate utilization in fish.22 The present work, therefore, seeks to address this gap by evaluating the potential of TPS as a functional supplement to enhance carbohydrate utilization in aquaculture species.
While TPS has been shown to exert antihyperglycemic effects in mammals, including the inhibition of intestinal glucose absorption,23,24 improvement of insulin sensitivity,25 and reduction of blood glucose levels,26,27 its relevance to fish remains uncertain. This uncertainty stems from fundamental differences in glucose regulation and postprandial metabolism between mammals and teleosts. In particular, glucose homeostasis in fish is governed more directly by hepatic nutrient sensing and substrate-level regulation than by the insulin-dependent signaling dominant in mammals.5,28,29 Consequently, it is unclear whether dietary TPS can alleviate carbohydrate-induced metabolic stress or enhance carbohydrate utilization in fish under high-carbohydrate feeding regimes. To address this gap, the present study mechanistically tests the hypothesis that TPS modulates key hepatic pathways of glucose metabolism in teleosts. In the process of glucose metabolism, key molecular components include the facilitative glucose transporter GLUT2, the glycolytic regulatory enzymes glucokinase (GCK) and pyruvate kinase (PK), as well as the rate-limiting enzymes for glycogen synthesis and degradation, glycogen synthase (GYS) and glycogen phosphorylase (PYGL), respectively.30 This targeted approach enables a focused evaluation of whether TPS influences major hepatic routes of postprandial glucose disposal in fish. Given the prominent role of hepatic substrate handling in teleosts, these endpoints provide a functionally relevant framework for assessing the metabolic efficacy of dietary TPS.
As a typical herbivorous cyprinid widely cultivated in aquaculture, grass carp (Ctenopharyngodon idella) demonstrates considerable glucose tolerance.31 However, the carbohydrate content in common commercial diets is frequently higher than its physiologically optimal level. Therefore, investigating exogenous additives to improve carbohydrate utilization in this widely cultured fish is particularly meaningful. In the present study, the effects of TPS on the growth performance and carbohydrate utilization of grass carp were evaluated. The findings are expected to provide a theoretical basis for the application of TPS to enhance carbohydrate utilization in aquatic animal feeds.
Materials and Methods
Experimental diets
The ingredients and chemical composition of the three experimental diets are presented in Table 1. Two basal diets were formulated with distinct levels of digestible carbohydrates—designated as the control and high-carbohydrate (HG) diets—using fish meal, casein, and gelatin as dietary protein sources; corn starch and α-starch as the primary sources of digestible carbohydrates; fish oil and soybean oil as lipid sources. The HG+TPS diet was formulated by supplementing the HG diet with TPS (TPS purity ≥ 98%; supplied by Wuhu Tianyuan Science & Technology Development Co., Ltd) at a concentration of 0.5 mg/g. The supplementation dose was determined according to previous studies on common carp.32 All raw materials were sourced from commercial suppliers in China.
After thorough blending of the ingredients, the mixtures were pelleted into 2.0 mm diameter pellets using a laboratory-scale pelletizer within 30 minutes. The pellets were subsequently dried in a precision forced-air drying oven (DGT-G-C series, Hefei Darth-Carter Scientific Equipment Co. Ltd) at 65 °C. Finally, the dried pellets were portioned into ziplock bags and stored at –20 °C until further use.
Fish and experimental conditions
Grass carp were obtained from a commercial fishery (Daxing, Hefei, China). Healthy, uniformly sized fish were randomly allocated into nine gray plastic tanks (60 × 60 × 60 cm; 180 L water volume), with 30 fish per tank. The tanks were then randomly assigned to one of three dietary treatments (control, HG, or HG+TPS), yielding three replicate tanks per treatment. Following a two-week acclimation period, during which the fish were fed a commercial diet (35.7% protein, 5.02% fat, 6.45% ash; Anhui Jinlong Feed Co., Ltd) twice daily to satiation, they were fasted for 24 hours to obtain initial body weights. The experiment commenced with 30 fish per tank, hand-fed twice per day (09:00 and 17:00) for eight weeks. Uneaten feed was collected via siphon 30 minutes after feeding, dried at 65 °C, and weighed to determine daily consumption. Throughout the trial, water temperature was maintained at 25.8 ± 0.4 °C, dissolved oxygen at 86.12 ± 2.89% saturation, water pH at 7.2-7.5, ammonia-nitrogen ≤ 0.05 mg/L, nitrite-nitrogen ≤ 0.01 mg/L, and photoperiod at 12L:12D.
Sample collection
At the end of the feeding trial, fish were fasted for 24 h, anesthetized with MS-222 (200 mg L⁻¹; Sigma-Aldrich, Germany), and weighed. Blood samples were collected from the caudal vein of four fish per tank using heparinized syringes for glucose measurement. Liver, muscle, and intestinal tissues were then excised, snap-frozen in liquid nitrogen, and stored at −80 °C for further analysis.
Chemical analyses
Diet composition was determined following standard methods (AOAC, 1995). Crude protein (N × 6.25) was measured using the Kjeldahl method with a Kjeltec system (K9860, Hanon Instruments, China). Crude lipid was quantified by ether extraction using a Soxtec System HT (HT6, Tecator, Sweden). Carbohydrate content was determined using the 3,5-dinitrosalicylic acid method. Crude ash was measured by combustion at 550 °C for 12 h in a muffle furnace (SX2-4-10, Zhengda Electric Technology Co., China). Gross energy was determined via bomb calorimetry (Parr 6200, Parr Instrument Company, USA).
Blood glucose levels, glycogen content detection and intestinal digestive activity analysis
Blood glucose levels were immediately measured using a handheld glucometer (Roche, Shanghai Rongsheng Bio-Pharmaceutical Co., China). Liver and muscle tissues (0.2 g) were homogenized in physiological saline and centrifuged (12,000× g, 10 min, 4 °C). Glycogen content was determined using a commercial Glycogen Assay Kit (A043-1-1, Nanjing Jiancheng Bioengineering Institute, China). Intestinal samples (0.1 g) were homogenized and centrifuged (6,000× g, 10 min, 4 °C), and the supernatants were used to assess α-amylase and α-glucosidase activities with respective assay kits (BC0610 and BC2550, Solarbio, China).
Gene expression assay
Total RNA was extracted from liver samples using Trizol reagent (Takara, Japan) and quantified spectrophotometrically (NanoDrop 2000, Thermo Scientific, USA). RNA purity was confirmed by A260/A280 ratios ≥ 2.0. cDNA was synthesized from 1 µg of RNA using SuperScript™ II RT reverse transcriptase (Takara). The primers for glucose metabolism-related genes (gys, pygl, glk, pk, glut2) were designed using Primer Premier 5.0 software and synthesized by Shanghai Sangon Biotech Co., Ltd. The specific criteria were: length of 18 - 25 base pairs, GC content of 45 - 55%, and melting temperature (Tm) of 55 – 65 °C. Primer specificity was preliminarily evaluated by performing BLASTn searches against the grass carp genome database to confirm unique target alignment. After synthesis, their specificity was further confirmed by 1.5% agarose gel electrophoresis (a single clear band) and melting curve analysis in qPCR (a single sharp peak). Quantitative real-time PCR was performed with gene-specific primers (Table 2) on a CFX Connect system (Bio-Rad, USA). Each 20 µL reaction contained 10 µL of 2× SYBR Green Master Mix (Vazyme, China), 0.8 µL of primers (10 µM each), 1 µL of cDNA (50 ng/µL), and 8.2 µL of ddH₂O. The thermal profile included initial denaturation (95 °C, 5 min), 40 cycles of amplification (95 °C for 10 s, Tm for 30 s), and a melt curve analysis (65 – 95 °C, increment 0.5 °C every 6 s). Two reference genes (β-actin and ef1α) were used for normalization. Gene expression was calculated via the 2^(–ΔΔCt) method, with all reactions run in triplicate.
Statistical analysis
Data are expressed as mean ± standard error of the mean (SEM). The tank was defined as the unit of biological replication for all statistical analyses. Prior to conducting analysis of variance (ANOVA), the normality of the data distribution was assessed using the Shapiro–Wilk test, and the homogeneity of variances across groups was verified via Levene’s test. Since the core assumptions of ANOVA were fully satisfied, no data transformation was performed for the subsequent statistical analysis. One-way ANOVA followed by Duncan’s multiple range test was applied to evaluate the significant differences in measured indices among dietary treatments. All statistical analyses were conducted using the Statistical Product and Service Solutions (SPSS) software (Version 18.0), and statistical significance was set at p < 0.05 for all tests.
Results
The effects of dietary TPS supplementation on growth performance and feed utilization
As summarized in Table 3, the inclusion of TPS in the diet notably influenced growth performance and feed utilization. At the end of the trial, final weight (FW), feed intake (FI), and specific growth rate (SGR) were significantly lower in the HG and HG+TPS groups relative to the control group (p < 0.05). However, supplementation with TPS significantly improved both FW and SGR compared to the HG group (p < 0.05). In addition, feed efficiency (FE) and protein efficiency (PE) were markedly reduced in fish fed the HG diet when compared to the control group (p < 0.05).
The effects of dietary TPS supplementation on blood glucose and glycogen contents
As illustrated in Figure 1, dietary TPS administration significantly modulated circulating glucose and hepatic glycogen levels. Blood glucose levels were significantly elevated in the HG group compared to the control group (p < 0.05). In contrast, fish receiving the HG+TPS diet exhibited a pronounced increase in hepatic glycogen content relative to controls (p < 0.05). No statistically significant differences were detected in muscular glycogen content across the experimental groups.
The effects of dietary TPS supplementation on the activities of carbohydrate hydrolases
The effects of TPS on intestinal carbohydrate-digesting enzyme activities are presented in Figure 2. No significant differences were observed in α-amylase activity among the control, HG, and HG+TPS groups. However, dietary inclusion of TPS significantly reduced intestinal α-glucosidase activity compared to the HG group (p < 0.05).
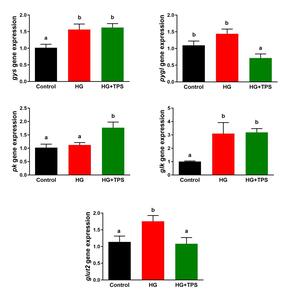
The effects of dietary TPS supplementation on the expressions of genes related to glucose metabolism
Figure 3 displays the mRNA expression levels of key genes involved in hepatic glucose metabolism. The expression of gys was significantly upregulated in both the HG and HG+TPS groups relative to the control (p < 0.05). In the HG+TPS group, pygl transcript levels were significantly downregulated compared to both the control and HG groups (p < 0.05). With respect to glycolysis, pk mRNA abundance was significantly higher in the HG+TPS group than in both the control and HG groups (p < 0.05). The glk gene expression was significantly enhanced in the HG and HG+TPS groups compared to the control (p < 0.05). Moreover, glut2 expression was significantly upregulated in the HG group relative to both the control and HG+TPS groups (p < 0.05).
Discussion
Increasing the efficiency of carbohydrate utilization to meet energy needs and spare expensive dietary nutrients is a viable nutritional strategy in the aquafeed industry. To explore suitable exogenous additives for enhancing carbohydrate utilization, we focused on natural tea extracts that have been shown to reduce blood glucose levels in mammals. Previous studies reported that oral treatment with TPS could improve growth performance in rats, including anti-obesity effects 33,.33 In our study, we supplemented TPS into a high-carbohydrate diet to estimate its effects on growth performance and carbohydrate utilization in grass carp. In the absence of supplemented diets, improved growth performance and protein-sparing effects were observed in experimental fish after TPS supplementation under high-carbohydrate diet intake — a finding reported for the first time in grass carp.
Our findings demonstrate that grass carp fed the HG diet exhibited a significant decrease in growth performance compared to the control group, a pattern consistent with previous reports of growth suppression due to excessive carbohydrate intake in grass carp,34 Tilapia nilotica (Oreochromis niloticus),35 and juvenile spotted Babylon (Babylonia areolata).36 In contrast, TPS supplementation significantly improved growth performance. This improvement coincided with enhanced postprandial glucose handling and hepatic glucose disposal in our study, indicating that TPS may alleviate carbohydrate-induced metabolic stress through coordinated metabolic regulation. Specifically, it was observed that TPS supplementation induced glycolysis by upregulating hepatic glk transcriptional levels, a key adaptive response to high carbohydrate intake, as grass carp typically enhance glycolysis and stimulate glucose-induced lipogenesis to manage glucose metabolism.37 Furthermore, glucose storage as hepatic glycogen was promoted through upregulated gys expression. This mechanism aligns with findings in rainbow trout hepatocytes, where the balance between glycogen phosphorylase and synthase determines hepatic glycogen accumulation.38 Therefore, as previously reported in teleosts, the strategy adopted by our experimental fish for glucose metabolism in response to higher dietary carbohydrate intake is to activate hepatic glycolysis and glycogen synthesis.
Many studies have demonstrated that suppressing gastrointestinal enzymes involved in nutrient digestion, particularly α-amylase and α-glucosidase activities, is one strategy to modulate glucose absorption following TPS administration in mammals.39,40 This effect is attributed in part to the ability of TPS to form complexes with α-amylase under simulated intestinal conditions, thereby inhibiting its activity.41 However, findings from mammalian systems cannot be directly extrapolated to teleosts, which may exhibit distinct digestive physiology and enzyme characteristics. For example, α-amylase in Nile tilapia (Oreochromis niloticus) possesses unique structural features and maintains activity across a broad pH range, suggesting potential species-specific functional resilience.42 In the present study, we observed no significant difference in intestinal α-amylase activity between grass carp fed the HG diet with or without TPS supplementation. This result indicates that, under our experimental conditions, TPS did not measurably affect starch hydrolysis via α-amylase inhibition. The mechanism underlying this absence of effect remains unclear and warrants further investigation through enzyme kinetics, binding assays, or structural studies using fish-specific α-amylase. In contrast, intestinal α-glucosidase activity was significantly reduced in TPS-supplemented fish. In teleosts, α-glucosidase is a key terminal digestive enzyme that catalyzes the hydrolysis of dietary oligo- and disaccharides into absorbable glucose, thereby directly shaping postprandial glucose influx and glycemic load.43 Accordingly, the marked reduction in α-glucosidase activity observed here provides fish-specific evidence that TPS can attenuate terminal carbohydrate digestion and limit glucose availability for absorption after feeding, which likely contributes to alleviating glucose stress under a high-carbohydrate diet. Together, these results suggest that TPS preferentially targets terminal carbohydrate digestion in grass carp, highlighting a functionally important route by which dietary TPS may modulate postprandial glucose dynamics in aquaculture species.
To further explore the molecular mechanism by which TPS regulates blood glucose in grass carp, we examined the expression of key rate-limiting genes involved in hepatic glucose metabolism. Our results showed that, compared to the HG group, TPS supplementation significantly upregulated the expression of hepatic pk, thereby stimulating glycolysis. Concurrently, TPS significantly downregulated the gene expression of pygl, while the expression of gys showed no significant change. This pattern suggests that in grass carp, TPS may promote net hepatic glycogen accumulation primarily by inhibiting glycogen breakdown rather than directly enhancing its synthesis. It is noteworthy that this strategy of promoting glycogen storage by downregulating pygl aligns with metabolic adaptive strategies employed by other teleosts to manage glucose.44 However, the specific molecular pathways may exhibit species-specific differences. For instance, studies in mammals have reported that black tea can increase hepatic glycogen content by upregulating gys and downregulating glycogen synthase kinase-3β (GSK-3β).45 The absence of significant gys upregulation observed in our grass carp study suggests that the terminal effectors of TPS-mediated glycogen metabolism regulation may differ between fish and mammals. Furthermore, dietary TPS supplementation in our study also reduced the gene expression of the hepatic glucose transporter 2 (glut2). This observation coincides with the previously noted significant decrease in intestinal α-glucosidase activity, jointly indicating that TPS can effectively mitigate postprandial glucose load through a dual mechanism: inhibiting exogenous glucose absorption in the intestine and reducing hepatic uptake of circulating glucose45,.46 Therefore, the improved SGR in the TPS group is consistent with a coordinated metabolic adjustment that may alleviate glucose overload and improve nutrient utilization under high-carbohydrate feeding. Specifically, TPS supplementation reduced glucose influx by inhibiting intestinal α-glucosidase and downregulating hepatic glut2, while simultaneously promoting hepatic glucose disposal through upregulated pk and downregulated pygl to stimulate glycolysis and glycogen storage. While previous epidemiological studies have reported similar hypoglycemic effects of green tea extract or TPS in diabetic rats48,,47 our study provides evidence in grass carp, identifying key targets across digestion, transport, and hepatic metabolism, and elucidating a distinct, species-specific pathway of TPS action. However, given that our study relied on selected molecular and biochemical endpoints, future studies incorporating enzyme kinetics, metabolic flux analysis, or targeted interventions are needed to establish definitive causality.
In summary, the results show that TPS supplementation improves the growth performance of grass carp fed a high-carbohydrate diet and promotes blood glucose clearance by enhancing glycogen synthesis and glycolysis while inhibiting intestinal α-glucosidase activity. The application of TPS shows potential as a natural antihyperglycemic feed additive to improve carbohydrate utilization, thereby providing benefits including dietary protein-sparing and cost savings in aquaculture.
Acknowledgments
The present study was supported by Natural Science Research Project of Anhui Educational Committee (2023AH051033, 2023AH051026), Anhui Provincial Natural Science Foundation (2308085Y26, 2308085QC109), and Research Funds of Joint Research Center for Food Nutrition and Health of IHM (2023SJY01).
Authors’ Contribution
Methodology: Linghong Wang (Lead). Formal Analysis: Linghong Wang (Lead). Investigation: Linghong Wang (Lead). Writing – original draft: Linghong Wang (Lead). Writing – review & editing: Shan Luo (Equal), Jinqian Huang (Equal). Supervision: Shan Luo (Equal), Jinqian Huang (Equal). Resources: Shengzhen Jin (Equal), Yanpeng Zhang (Equal). Conceptualization: Xiaochen Yuan (Lead). Funding acquisition: Xiaochen Yuan (Lead).
Competing of Interest – COPE
No competing interests were disclosed.
Ethical Conduct Approval – IACUC
The animals and experiments were conducted in compliance with the “Guidelines for Experimental Animals” of the Ministry of Science and Technology (Beijing, China). The study was approved by the Institutional Animal Care and Use Ethics Committee of Anhui Agricultural University. All efforts were made to minimize suffering.
Informed Consent Statement
All authors and institutions have confirmed this manuscript for publication.
Data Availability Statement
All data are available upon reasonable request.

__hepatic_glycogen_(b)_and_musc.jpeg)
_and_-glucosidase_(b)_activities_in_intest.jpeg)
__hepatic_glycogen_(b)_and_musc.jpeg)
_and_-glucosidase_(b)_activities_in_intest.jpeg)